D block elements are the elements that can be found from the third group to the twelfth group of the modern periodic table. The valence electrons of these elements fall under the d orbital. D block elements are also referred to as transition elements or transition metals. The first three rows of the d block elements, which correspond to the 3d, 4d, and 5d orbitals, respectively, are given in the below article.
Download Complete Chapter Notes of The d & f – Block Elements
Download Now
Table of Contents
- Position
- D Block as Transition Element
- Electronic Configuration
- Atomic and Ionic Radii
- Properties
- Oxidation States
- Formation of Coloured Ions
- Alloy Formation
- Important Compounds
What Are D Block Elements?
Elements having electrons (1 to 10) present in the d-orbital of the penultimate energy level and in the outermost ‘s’ orbital (1-2) are d block elements. Although electrons do not fill up ‘d’ orbital in the group 12 metals, their chemistry is similar in many ways to that of the preceding groups, and so considered as d block elements.
These elements typically display metallic qualities such as malleability and ductility, high values of electrical conductivity and thermal conductivity and good tensile strength. There are four series in the d block corresponding to the filling up of 3d, 4d, 5d or 6d orbitals.
- 3d- Sc, Ti, V, Cr, Mn, Fe, Co, Ni, Cu, Zn
- 4d- Y, Zr, Nb, Mo, Tc, Ru, Rh, Pd, Ag, Cd
- 5d- La, Hf, Ta, W, Re, Os, Ir, Pt, Au, Hg
- 6d- incomplete.
There are 10 elements filling up the ‘d’ orbital in each series.
⇒ Also Read
Position of D Block Elements in Periodic Table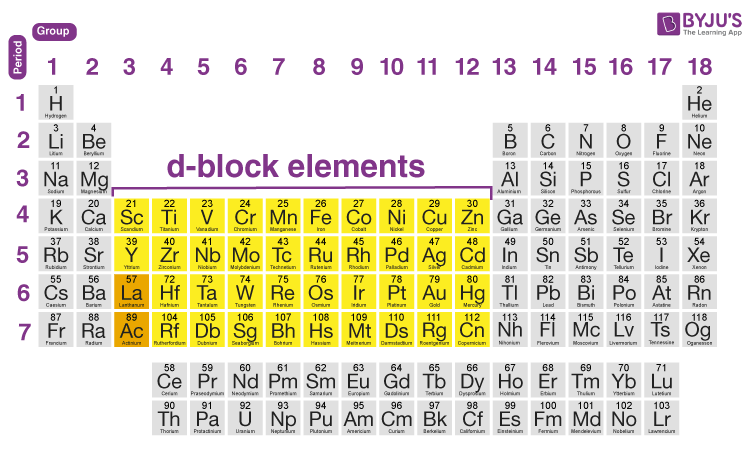
D block elements occupy columns 3 to 12 and may have atoms of elements with completely filled ‘d’ orbital. IUPAC defines a transition metal as “an element whose atom or its cations has a partially filled d sub-shell.
Why are D Block Elements called Transition Elements?
Transition elements occupy groups 4-11. Scandium and yttrium of group 3, having a partially filled d subshell in the metallic state, also are considered as transition elements. Elements like Zn, Cd and Hg of the 12 columns of the d block have completely filled the d-orbital and hence are not considered as transition elements.
Transition elements are so-called, indicating their positioning and transition of properties between s and p block elements. So, all the transition metals are d block elements, but all the d block elements are not transition elements.
Properties of Transition Metals
- Electrons are added to the ‘d’ sub-orbitals that lie between their (n+1) s and (n+1) p sub-orbitals.
- Placed between s and p block elements in the periodic table.
- Properties between s and p-block elements.
Electronic Configuration of D Block Elements
D block Elements have a general electronic configuration of (n-1)d 1-10ns 1-2. These elements can find stability in half-filled orbitals and completely filled d orbitals. An example of this will be the electronic configuration of chromium, which has half-filled d and s orbitals in its configuration – 3d54s1. The electronic configuration of copper is another example. Copper displays an electronic configuration of 3d104s1 and not 3d94s2.
This can be attributed to the relative stability of the completely filled d orbital. Zinc, Mercury, Cadmium, and Copernicium exhibit completely filled orbitals in their ground states as well as in their general oxidation states. For this reason, these metals are not considered as transition elements, whereas the others are d block elements.
- The electronic configuration for period 4, transition elements is (Ar) 4s 1-2 3d 1-10
- The electronic configuration for period 5, transition elements is (Kr) 5s 1-2 4d 1-10
- The electronic configuration for period 6, transition elements is (Xe) 4s 1-2 3d 1-10
Along the period, from left to right, electrons are added to the 3d subshell as per the Aufbau principle and Hund’s rule of multiplicity.
| 1st transition series | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn |
| 4s23d1 | 4s23d2 | 4s23d3 | 4s13d5 | 4s23d5 | 4s23d6 | 4s23d7 | 4s23d8 | 4s13d10 | 4s23d10 | |
| 2nd transition series | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd |
| 5s24d1 | 5s24d2 | 5s14d4 | 5s14d5 | 5s24d5 | 5s14d7 | 5s14d8 | 5s04d10 | 5s14d10 | 5s24d10 | |
| 3rd transition series | La | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg |
| 6s25d1 | 6s25d2 | 6s25d3 | 6s25d4 | 6s25d5 | 6s25d6 | 6s25d7 | 6s15d9 | 6s15d10 | 6s25d10 |
Anomalies do occur in all the series, which can be explained by the following considerations.
- The energy gap between the ns and (n-1) d orbitals
- Pairing energy for the electrons in s-orbital
- Stability of half-filled orbitals to the partly-filled orbitals.
Chromium has a 4s13d5 electron configuration rather than the 4s23d4 configuration, and copper has 4s13d10 rather than 4s23d9. These anomalies in the first transition series can be understood from the stability of half-filled orbitals compared to the partly-filled orbitals.
In the second series, transition metals from niobium, electron presence in d orbitals appear to be preferred than being shared in s orbitals. Between the available s and d orbitals, the electron can either go for sharing in the s-orbital or excited to the d-orbital. Obviously, the choice depends on the repulsive energy it has overcome on sharing and the energy gap between the s and d-orbitals.
In the second series, s and d-orbital have almost the same energy, because of which electrons prefer to occupy the d-orbital. So, from niobium, the s-orbital has mostly only one electron. Third-series transition metals, on the other hand, have more paired s configurations even at the expense of half-filled orbitals (Tungsten- 6s25d4). This series comes after the filling up of 4f orbitals and resulting lanthanide contraction.
The reduced size results in the high shielding of d orbitals by the ‘f’ electron. This shielding increases the energy gap between the s and 5d orbitals such that pairing energy is less than the excitation. Excitation of the electron does not take place in tungsten, in spite of the stability possible because of half-filled orbitals.
Atomic and Ionic Radii of D Block Elements

Metallic Radii of 1st, 2nd, and 3rd Row Transition Metals
Atomic and ionic radii of elements of all three-transition series
- Decreases rapidly, from columns 3 to 6
- Remains steady from columns 7 to 10 and
- Starts increasing from columns 11 to 12.
For example, in the first transition series, atomic radii, the decrease is more from Sc to Cr (group 3 to 6 ), and is almost the same for Mn, Fe, Co, and Ni (groups 7, 8, 9 & 10) and increase in Cu and Zn.
- The larger decrease in atomic radii, in columns 3 to 6 elements, is due to the increase in effective nuclear charge but poor shielding because of the smaller number of d-electrons.
- In elements of columns 7 to 10, increasing effective nuclear charge is balanced by the repulsion between the shared d electrons so that radii remain the same.
- In the case of 11 and 12 columns elements, the d orbital is full with ten electrons and shields the electrons present in the higher s-orbital. So, groups 11 and 12 elements like Cu and Zn have bigger sizes than their earlier elements in the block.
Since electrons occupy a higher orbital, the radii of the third series are to be more than the second series elements. But the radii of both series are almost the same. In the third series of elements, 5d orbitals are filled only after the filling up of 4f orbitals, which increases the effective nuclear charge by 14 units.
This higher nuclear charge leads to the larger shrinkage of radii, known as Lanthanide contraction. An increase in radii due to the higher orbital will be effectively neutralised by the increase in the effective nuclear charge. So, the radii of the second and third series elements have the same atomic radii. For example, Niobium and hafnium have almost the same atomic radii.
Properties of D Block Elements
Ionization Energy of D Block Elements
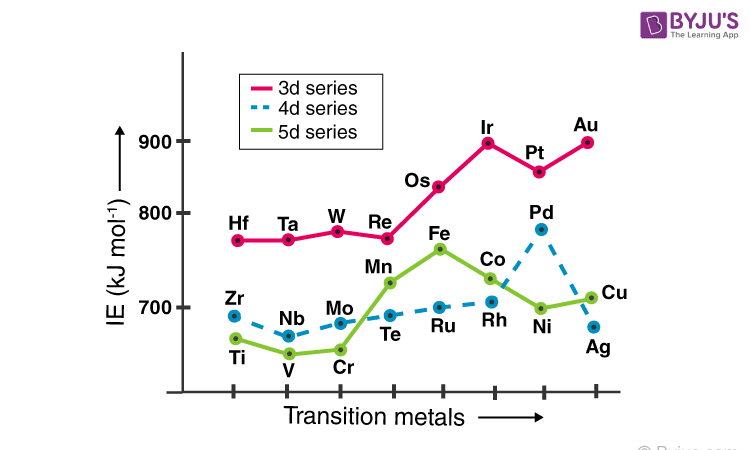 Ionization energy is the energy needed to remove the valence electron from the atom/ion and is directly related to the force of attraction on the electron. Hence, the larger the nuclear charge and the smaller the radii of the electron larger will be the ionization energy (IE). Ionization energy also will be more for half-filled and fully-filled orbitals.
Ionization energy is the energy needed to remove the valence electron from the atom/ion and is directly related to the force of attraction on the electron. Hence, the larger the nuclear charge and the smaller the radii of the electron larger will be the ionization energy (IE). Ionization energy also will be more for half-filled and fully-filled orbitals.
The Ionization energy of the d block elements is larger than s-block and smaller than the p-block elements between which they are placed. In the first series, except for chromium and copper, the first ionization energy involves removal from the filled s-orbital. Among them, the ionization energy of d block elements increases with the increase in atomic number up to Fe.
In Co and Ni, increasing sharing of d-electrons compensates for the atomic number increase resulting in the decrease of ionization energy. Copper and zinc show increasing IE as s -block elements. In the second series, elements from Niobium have single electrons in the s-orbital.
Hence, they show a gradual increase in IE with increasing atomic number. Palladium, on the other hand, has a completed d-shell and no electron in the s-shell. So, Pd shows the maximum IE. Because of lanthanide contraction, the attraction of electrons by the nuclear charge is much higher, and hence IE of 5d elements are much larger than 4d and 3d. In the 5d series, all elements except Pt and Au have filled the s-shell.
Elements from Hafnium to rhenium have the same IE, and after IE increases with the number of shared d-electrons such that Iridium and Gold have the maximum IE.
Metallic Character
D block elements show typical metallic behaviour of high tensile strength, malleability, ductility, electrical and thermal conductivity, metallic lustre, and crystallise in bcc/ccp/hcp structures.
They are very hard and have high enthalpy of atomization and low volatility, except for copper. Hardness increases with the number of unpaired electrons. Hence, Cr, Mo and W are very hard metals among d block elements. The group-12 elements (Zn, Cd and Hg) show the exception in this regard also.
Oxidation States of D Block Elements
The oxidation state is a hypothetical state where the atom appears to release or gain more electrons than the usual valency state. It is still useful in explaining the properties of the atom/ion. Transition elements/ions may have electrons in both s and d-orbitals.
Since the energy difference between s and d-orbital is small, both the electrons can involve in ionic and covalent bond formation, and hence exhibit multiple (variable) valency states (oxidation states).
Each transition element can hence exhibit a minimum oxidation state corresponding to the number of s-electrons and a maximum oxidation state equivalent to the total number of electrons available in both s and d-orbitals. In between, oxidation states also become possible.
| Sc | +2,+3 | +3 | +3 | Y | +2,+3 | +3 | +3 | La | +2,+3 | +3 | +3 | |
| Ti | +2,+3,+4, | +2 | +4 | Zr | +2,+3,+4, | +2 | +4 | Hf | +2,+3,+4, | +4 | +4 | |
| V | +2,+3,+4,+5, | +2 | +5 | Nb | +2,+3,+4,+5, | +2 | +5 | Ta | +2,+3,+4,+5, | +4 | +5 | |
| Cr | +2,+3,+4,+5,+6, | +1 | +2 | +6 | Mo | +2,+3,+4,+5,+6, | +4 | +6 | W | +2,+3,+4,+5,+6, | +4 | +6 |
| Mn | +2,+3,+4,+5,+6,+7 | +2 | +7 | Tc | +2,+3,+4,+5,+6,+7 | +4 | +7 | Re | +2,+3,+4,+5,+6,+7 | +4 | +7 | |
| Fe | +2,+3,+4,+5,+6, | +2 | +6 | Ru | +2,+3,+4,+5,+6,+7,+8 | +4 | +8 | Os | +2,+3,+4,+5,+6,+7,+8 | +4 | +8 | |
| Co | +2,+3,+4, | +2 | +4 | Rh | +2,+3,+4, | +3 | +4 | Ir | +2,+3,+4, | +4 | +4 | |
| Ni | +2,+3,+4, | +2 | +4 | Pd | +2,+3,+4, | +2 | +4 | Pt | +2,+3,+4, | +4 | +4 | |
| Cu | +1,+2, | +1 | +2 | +2 | Ag | +1,+2, | +1 | +2 | Au | +1,+2, | +1 | +2 |
| Zn | +2, | +2 | +2 | Cd | +2, | +2 | +2 | Hg | +2, | +1 | +2 | +2 |
Trends in the Oxidation States
1. The minimum Oxidation state of 1 is shown by Cr, Cu, Ag, Au and Hg.
2. More stable oxidation state increases in the order 3d ˂ 4d ˂ 5d. 3d series elements are most stable in +2; 4d series in +2 and +4 and 5d series in +4. Cr6+ and Mn7+ (of 3d) are not stable in their higher OS. Compounds containing them, CrO42- and MnO4– are very reactive and strong oxidizing agents.
While Mo6+ and Tc7+ (of 4d) are stable in their higher OS, compounds containing them, MoO42- and TcO4–, are unreactive and stable. Similarly, W6+ and Re7+ (of 5d) are stable in their higher OS. Compounds containing them, WO42- and ReO4– are unreactive and stable.
Cations of the second and third-row transition metals in lower oxidation states (+2 and +3) are much more easily oxidized than the corresponding ions of the first-row transition metals. For example, the most stable compounds of chromium are those of Cr(III), but the corresponding Mo(III) and W(III) compounds are highly reactive.
In fact, they are often pyrophoric, bursting into flames on contact with atmospheric oxygen. As we shall see, the heavier elements in each group form stable compounds in higher oxidation states that have no analogues with the lightest member of the group.
3. Strongly oxidizing, high oxidation number elements form compounds of oxides and fluorides and not bromides and iodides.
Vanadium form only VO4–, CrO42-, MnO4–, VF5, VCl5, VBr3, VI3 and not VBr5, VI5. V5+ oxidizes Br– and I– to Br2 and I2 but not fluoride because of its high electronegativity and small size.
Similarly, strongly reducing, low oxidation number elements form bromides and iodides and not oxides and fluorides.
4. Maximum oxidation state equal to the s and d-electrons is exhibited by middle-order elements in each series. Thus, manganese in the 3d series has +7, Ru in 4d and Os in 5d possess +8 maximum oxidation state.
5. Elements may show all the oxidation states between the minimum and maximum.
6. Elements in their lower oxidation states will be ionic and basic (TiO, VO, CrO, MnO, TiCl2 and VCl2) in-between state amphoteric (Ti2O3, V2O3, Mn2O3, CrO3, Cr2O3, TiCl3, VCl3 ) and higher oxidation state will be covalent and acidic (V2O5, MnO3, Mn2O7, VCl4 and VOCl3 ).
7. Lower oxidation state may get stabilised by back bonding in complexes, Ni(CO)4, Fe(CO)5, [Ag(CN)2]–, [Ag(NH3)2]+
Lower oxidation states in these metals are stabilised by ligands like CO, which are pi-electron donors, whereas the higher oxidation states are stabilised by electronegative elements like Fluorine(F) and Oxygen(O). Hence, the high oxidation compounds of these metals are mainly fluorides and oxides.
8. Relative stabilities of the oxidation states depend on many factors, like, the stability of the resulting orbital, IE, electronegativity, enthalpy of atomization, enthalpy of hydration, etc.
- Ti4+ (3d0) is more stable than Ti3+(3d1). Mn2+ (3d5) is more stable than Mn3+(3d4).
- Ionization energies contribute to the relative stability of transition metal compounds (ions). For example, Ni2+ compounds are thermodynamically more stable than Pt2+, whereas, Pt4+ compounds are more stable than Ni4+. The relative stabilities can be explained as follows:
| Metal | (IE1+IE2) kJmol−1 | (IE3+IE4) kJmol−1 | Etotal =(=IE1+IE2+IE3+IE4) kJ mol−1 |
| Ni | 2490 | 8800 | 11290 |
| Pt | 2660 | 6700 | 9360 |
Thus, the ionization of Ni to Ni2+ requires lesser energy (2490 kJ mol−1) as compared to the energy required for the production of Pt2+ (2660 kJ mol−1). Therefore, Ni2+ compounds are thermodynamically more stable than Pt2+ compounds.
On the other hand, the formation of Pt4+ requires lesser energy (9360 kJ mol1) as compared to that required for the formation of Ni4+ (11290 kJ/mol). Therefore, Pt4+ compounds are more stable than Ni4+ compounds. This is supported by the fact that [PtCl6]2+ complex ion is known, while the corresponding ion for nickel is not known.
9. In p-block, the heavier elements prefer lower oxidation states due to what is called the inert pair effect. But in the case of d block elements, the higher oxidation states are more stable for heavier members in a group.
Electrode Potential in D Block Elements
Relative stabilities of transition metal ions in different oxidation states in the aqueous medium can be predicted from the electrode potential data. The oxidation state of a cation for which ΔH(ΔHsub + lE + ΔHhyd) or E° is more negative (for less positive) will be more stable.
- E° becomes less negative along the series, indicating higher stability of the reduced state.
- Compared to first and second group metals, transition elements have low E°.
Physical Properties of d Block Elements
Density: Among the transition series, the trend in density will be the reverse of atomic radii, i.e., the density increase remains almost the same and then decreases along the period. 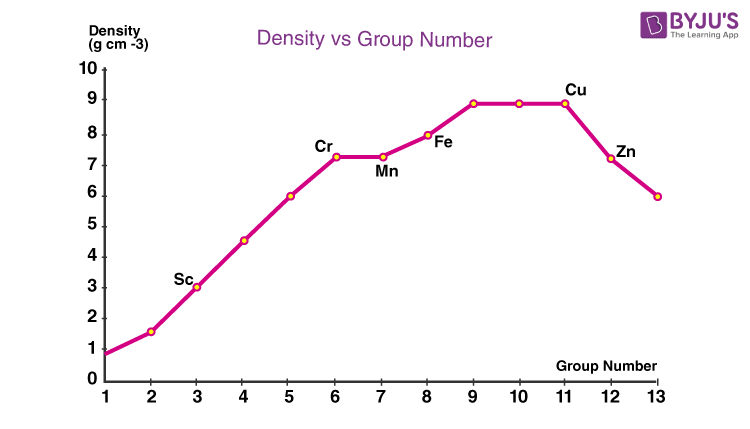
Down the column density of the 4d series is larger than 3d. Due to lanthanide contraction and a larger decrease in atomic radii, the volume density of 5d series transition elements is double than 4d series.
In the 3d series, scandium has the lowest density, and copper has the highest density. Osmium (d=22.57g cm-3) and iridium (d=22.61g cm-3) of the 5d series have the highest density among all d block elements.
Some relative radii of d block elements are Fe ˂ Ni ˂ Cu, Fe ˂ Cu ˂ Au, Fe ˂ Hg ˂ Au.
Why Do the d Block Elements Have High Melting and Boiling Points?
Unpaired electrons and the empty or partially filled d-orbitals form covalent bonding in addition to the metallic bonding by s-electrons. Because of such strong bonding, d-block elements have high melting and boiling points than s and p block elements. This trend goes till d5 configuration and then decreases as more electrons get paired in the d-orbital.
- Cr, Mo and W possess the highest melting at boiling point in their series of elements.
- Manganese (Mn) and Technetium (Tc) have half-filled configurations resulting in weak metallic bonding and abnormally low melting and boiling points.
- Group 12, Zn, Cd and Hg have no unpaired d-electrons, and hence no covalent bonding. Their melting and boiling points will be the lowest in their series.
Mercury – the liquid metal: Mercury is the only metal that exists in its liquid state at room temperature. 6s valence electrons of Mercury are more closely pulled by the nucleus (lanthanide contraction), such that outer s-electrons are less involved in metallic bonding.
What Transition Elements Are Considered Noble Metals?
In the three transition series,
- The ionization energies of elements increase very slowly across a given row
- From the left of the 3d series to the right corner, 5d transition elements, density, electronegativity, and electrical and thermal conductivities increase, while enthalpies of hydration of the metal cations decrease in magnitude.
This indicates that the transition metals become steadily less reactive and more “noble” in character. The relatively high ionization energies, increasing electronegativity, and decreasing low enthalpies of hydration make metals (Pt, Au) in the lower right corner of the d block so unreactive that they are often called the “noble metals.”
Magnetic Properties of d Block Elements
Materials are classified by their interaction with the magnetic field:
- Diamagnetic: if repelled
- Paramagnetic: if attracted
- Ferromagnetic: if it can retain the larger magnetic nature even in the absence of a magnetic field
Paired electrons cause diamagnetism. Unpaired electrons result in para-magnetism and are aligned together, and they produce ferromagnetism. D block elements and their ions exhibit this behaviour depending on the unpaired electrons.
Unpaired electrons contribute to the ‘orbital magnetic moment’ and ‘spin magnetic moment’. However, for the 3d series, the orbital angular moment is negligible, and the approximate spin-only magnetic moment is given by the formula:
µ = √[4s (s + 1)] = √[n (n + 1)] BM
Where ‘S’ is the total spin and ‘n’ is the number of unpaired electrons. Its unit is Bohr Magneton (BM). For higher d-series, the actual magnetic moment includes components from the orbital moment in addition to the spin moment. Chromium and molybdenum possess the maximum number (6) of unpaired electrons and magnetic moment.
| Ion | Outer Configuration | No. of Unpaired Electrons | Magnetic Moment (BM) | |
| Calculated | Observed | |||
| Sc3+ | 3d0 | 0 | 0 | 0 |
| Ti3+ | 3d1 | 1 | 1.73 | 1.75 |
| Ti2+ | 3d2 | 2 | 2.84 | 2.86 |
| V2+ | 3d3 | 3 | 3.87 | 3.86 |
| Cr2+ | 3d4 | 4 | 4.90 | 4.80 |
| Mn2+ | 3d5 | 5 | 5.92 | 5.95 |
| Fe2+ | 3d6 | 4 | 4.90 | 5.0-5.5 |
| Co2+ | 3d7 | 3 | 3.87 | 4.4-5.2 |
| Ni2+ | 3d8 | 2 | 2.84 | 2.9-3.4 |
| Cu2+ | 3d9 | 1 | 1.73 | 1.4-2.2 |
| Zn2+ | 3d10 | 0 | 0 | 0 |
Formation of Coloured Ions by d Block Elements
Compounds of d block elements have a variety of colours. When a frequency of light is absorbed, the light transmitted exhibits a colour complementary to the frequency absorbed. Transition element ions can absorb the frequency in the visible region to use it two ways and produce visible colour.
1. d-d Transition
One way is the excitation of an electron to a higher energy level. In transition element ions, the presence of d-electron and empty d-orbital shall result in colour formation. Also, valence electron excitation and de-excitation. This is called the d-d transition.
D-orbitals are generally degenerate and have the same energy. The presence of ligands that can form coordinate bonds with these ions remove the degeneracy and split them into two groups, namely eg and t2g d-orbitals. The energy difference (∆E) depends on the strength of the incoming ligand.
Electrons in the lower d-orbitals can be excited into the higher d-orbitals by absorbing energy in the visible region (λ=400-700nm) and transmitting (giving) a colour complementary to it.
For example, [Cu(H2O)6] 2+ ions absorb red radiation and appear complementary blue-green. Hydrated Co2+ ions absorb radiation in the blue-green region and, therefore, appear red in the sunlight.
Cupric ion is colourless, and in the presence of water, molecules become blue in colour.
a) Colour of the ions varies with their oxidation state. The Cr6+ in potassium dichromate is yellow in colour, whereas Cr3+ and Cr2+ are generally green and blue, respectively.
b) Colour of the compound is dependent on the complexing or coordinating group also. For example, Cu2+ shows light blue colour in the presence of water as the ligand but a deep blue colour in the presence of ammonia as the ligand.
c) Transition metal ions which have:
- Completely filled d-orbitals having no vacant d-orbitals for the excitation of electrons are colourless. Cu+(3d10), Zn2+(3d10), Cd2+(4d10), Hg2+(5d10), and Zn, Cd, and Hg are colourless.
- Transition metal ions which have completely empty d-orbitals without d-electrons are also colourless. Sc3++(3d0), and Ti4++(3d0), ions are colourless.
L-M and M-L dπ – pπ bonding
Ligands may donate their p electrons into the empty d orbitals of metal ions. This interaction, called ligand-metal / metal-ligand or dπ – pπ bonding, also may give colour to the compounds.
Complex Formation Tendency of D Block Elements
Complex compounds are compounds wherein a number of neutral molecules or anions are bound to a metal. Metals that are a part of the d block elements form many complex compounds owing to their small ionic size, high charge, and relative availability of d orbitals for the formation of bonds.
Transition metal and their ions, with their larger nuclear charge and smaller size, can attract electrons and receive lone pairs of electrons from anions and neutral molecules into their empty d-orbitals, forming coordinate bonding.
Transition elements thus form complex molecules with CO, NO, NH3, H2O, F–, Cl–, and CN–. Examples of transition metal complexes are, [Co(NH3) 6] 3+ [Cu(NH3)4] 2+, Y(H2O) 6]2+, [Fe(CN)6]4−, [FeF6] 3−, [Ni(CO)4],
Catalytic Activity of Elements
Catalysts are important for the industrial bulk production of many chemicals. Many d-block elements, as metals are in their ionic form, are being used as a catalyst in many chemical and biological reactions.
Iron in Haber’s process to make ammonia, vanadium pentoxide in the manufacture of sulphuric acid, titanium chloride as Zigler Natta catalyst in polymerization and Palladium chloride in the conversion of ethylene to acetaldehyde are some very important commercial catalytic processes involving d block metals.
Most transition elements act as good catalysts because of,
- The presence of vacant d-orbitals.
- The tendency to exhibit variable oxidation states.
- The tendency to form reaction intermediates with reactants.
- The presence of defects in their crystal lattices.
They take the reaction through a path of low activation energy by:
- Providing a large surface area for absorption and allowing sufficient time to react.
- May interact with the reactants through their empty orbitals.
- May actively interact by redox reaction through their multiple oxidation states.
Alloy Formation in d Block Elements
Atomic radii of the transition elements in any series are not much different from each other. As a result, they can very easily replace each other in the lattice and form solid solutions over an appreciable composition range. Such solid solutions are called alloys. Atoms within 15% of the difference in radii can form alloys.
Alloys are homogeneous solid solutions of two metals or metals with a non-metal. The alloys of transition metals are hard, and high metals have high melting as compared to the host metal.
Various steels are alloys of iron with metals such as chromium, vanadium, molybdenum, tungsten, manganese, etc. Some important alloys are:
Bronze – Cu (75-90%) + Sn (10-25%), Chromium steel – Cr (2-4% of Fe) Stainless steel- Cr (12-14% and Ni (2-4%) of Fe, Solder- Pb +Sn
Interstitial Compounds of d Block Elements
Transition metal has a void in its crystal lattice structure. Small non-metallic atoms and molecules like hydrogen, boron, carbon etc., can be trapped in the void during crystal structure formation, and they are called interstitial compounds. They are neither ionic nor covalent and non-stoichiometric, as in TiH1.7 and VH0.56.
Interstitial compounds have the following properties:
- Their melting points are very high.
- They are extremely hard.
- They have similar conductivity properties when compared to other metals
- They are unreactive and tend to be chemically inert.
Examples of interstitial compounds that are formed with transition metals are TiC, Mn4N, Fe3H, and TiH2.
Non-Stoichiometric Compounds
Transition metal compounds of different oxidation states may sometimes present together. They may be formed by solid structure defects or by prevalent conditions. But, this mixture behaves like a single compound.
This compound will not have any finite composition or structure. Non-stoichiometric is shown particularly while combining with group 16 (O, S, Se, Te) elements. Examples: Fe0.94O, Fe0.84O, VSe0.98, Se1.2.
Important Compounds of d Block Elements
The d block elements form some compounds of vital industrial importance. Some such compounds include:
K2Cr2O7 (Potassium Dichromate)
This compound is considered very important in the leather industry. It is also used as an oxidant in most azo compound preparation processes.
The structure of the dichromate ion is made up of two tetrahedra that share a single corner with a chromium-oxygen-chromium bond angle of 1260. Potassium dichromate is a strong oxidizing agent. Potassium dichromate is also used as a primary standard in the process of volumetric analysis.
KMnO4 (Potassium Permanganate)
The physical appearance of KMnO4 has an intense purple colour. It exhibits diamagnetic properties and also weak paramagnetic properties, which are reliant on the temperature.
The permanganate ion exhibits diamagnetism due to the absence of unpaired electrons in it. Potassium permanganate is also used as an oxidant in the preparation of various products in organic chemistry. It also finds use in the bleaching of cotton, silk, and wool. It can also be used for the decolourization of oils owing to its strong oxidizing ability.
D and f block Elements – Oxidation States

15 Important Questions on D-Block and F-Block Elements

Alloy Formation of d and f Block Elements

Colour of Ions in d and f Block Elements


Comments