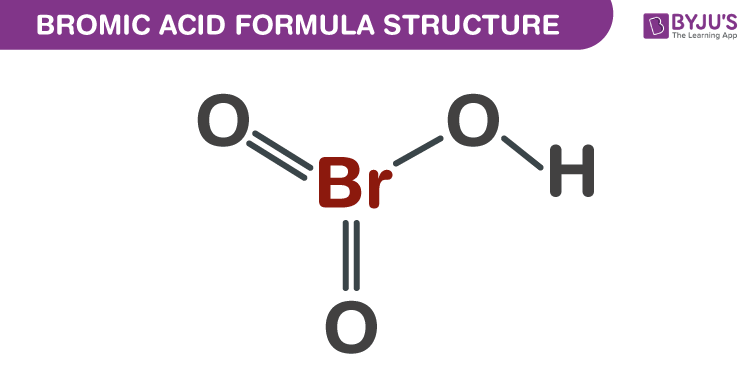
Bromic Acid formula, also known as Hydrogen bromate formula or Bromic (V) acid formula is explained in this article. It is an oxoacid and exists only in aqueous solution. It is a solution which is colourless. During its decomposition to bromine at room temperature, the colour of the solution changes yellow. The molecular or chemical formula of Bromic Acid is HBrO3.
Hydrogen bromate is a powerful oxidizing agent and is a common ingredient in Belousov-Zhabotinsky reaction. Bromic acid is obtained as a product in the reaction of barium bromate (Ba(BrO3)2) and sulfuric acid (H2SO4). Barium sulfate obtained is a precipitate form. Therefore bromic acid can be decanted by removing the precipitated barium sulfate.
Bromic Acid Formula Structure
Properties Of Bromic Acid
| Chemical formula | HBrO3 |
| Molecular weight | 128.91 g/mol |
| Conjugate base | Bromate |
| pKa | −2 |
| Complexity | 46.2 |
To learn more about Bromic Acid formula from the expert faculties at BYJU’S, register now! Also, you can download notes on Hydrogen bromate for free.
Comments