What is Calcium Carbonate?
Calcium carbonate is an inorganic chemical compound with the chemical formula CaCO3.
Calcium carbonate is one of the most popular chemicals which is first encountered in school classrooms, where the use of chalk (a form of CaCO3) is found. It is found in the earth’s crust. It is also found in many forms such as marble, limestone, etc. Although they are available in various forms they are chemically similar and only differ physically. They are also referred to as calcite.
Table of Contents
- Commercial Production
- Calcium Carbonate Structure
- Calcium Carbonate Formula
- Preparation of Calcium Carbonate
- Properties of Calcium Carbonate
- Application of Calcium Carbonate
- Calcium Carbonate Uses
- Frequently Asked Questions – FAQs
Calcium carbonate is a non-toxic and odourless compound commonly found as a white mineral which occurs naturally in chalks, limestones and marbles.
Commercial Production of Calcium Carbonate
Calcium carbonate is produced commercially in two different grades. Both grades compete industrially based primarily on particle size and the characteristics imparted to a product.
- Ground Calcium Carbonate – Produced via extraction and processing of naturally occurring deposits. GCC crystal shape is irregularly rhombohedral and has a broader size distribution.
- Precipitated Calcium Carbonate – Produced via chemical precipitation via a carbocation process or as a by-product of some bulk chemical processes. PCC crystal shape depends on the product and the particles are more uniform and regular with a narrow size distribution.
PCC has smaller particles has a higher purity is less abrasive and tends to have higher brightness than GCC.
Calcium Carbonate Structure
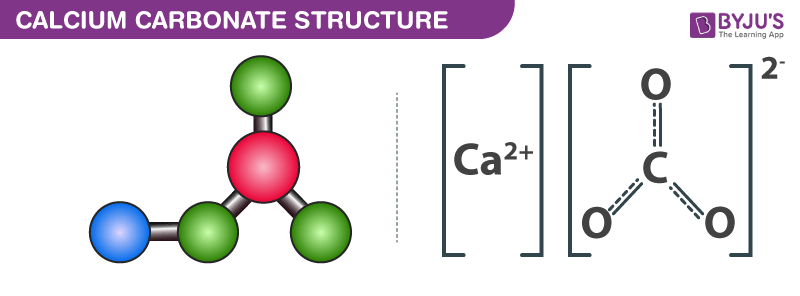
Structure of Calcium Carbonate
Calcium Carbonate Formula
- It is a chemical compound with the chemical formula CaCO3.
- It is a white insoluble powder-like substance which occurs naturally in minerals, chalk, marble, limestone, calcite, shells, pearl, etc.
- Medicinally, it is used as an antacid or as a calcium supplement. It is also used as filler in cosmetics. It is added to swimming pools as a disinfectant agent and a pH corrector.
- It finds extensive usage in the manufacturing industry as a building material (marble), ingredient for quick lime and cement.
Preparation of Calcium Carbonate – CaCO3
1. CaCO3 is obtained by using carbon dioxide and slaked lime as raw materials. When carbon dioxide is passed through slaked lime, calcite is obtained.
Another method to obtain calcite is by adding sodium carbonate to calcium chloride.
When carbon dioxide is passed in excess it leads to the formation of calcium hydrogen-carbonate.
2. On a large scale, it is prepared by passing carbon dioxide gas through calcium hydroxide (slaked lime). However, if carbon dioxide is passed in excess, it forms the soluble calcium hydrogen-carbonate.
Ca(OH)2 + CO2→ CaCO3 + H2O
Properties of Calcium Carbonate – CaCO3
- It is a fluffy powder.
- It decomposes to give carbon dioxide when heated up to 1200K.
- When it reacts with dilute acid, it liberates carbon dioxide as a by-product.
- At 1200K, calcium carbonate decomposes to give carbon dioxide and calcium oxide.
CaCO3 → CaO + CO2
- On reacting with dilute acids, calcium carbonate gives carbon dioxide.
CaCO3 + 2HCl → CaCl2 + H2O +CO2
Application of Calcium Carbonate
- Calcium carbonate is largely employed in the pulp and paper industry. It can be used as a filter and pigment, making possible the production of a whiter, higher quality pigment than other minerals.
- In the construction industry, calcium carbonate is used as a filler in concrete, increasing its durability and appearance and to purifying metals for use in construction applications.
- Another application of calcium carbonate is in fertilizers to provide calcium to plants and pH stabilization of the soil.
- Calcium carbonate can also be an additive to food products for livestock animals and humans and as a supplement in vitamins.
- In water and sewer treatment plants, calcium carbonate is employed in the removal of acidity and impurities.
Calcium Carbonate Uses
The list of uses of calcium carbonate is given below.
- It plays an important role in construction, be it as a building material (marble) or as an ingredient in cement.
- It is used in medicinal industries which manufacture antacids, tablets which are made of base materials etc.
- It is used as calcium supplement.
- It is used in the manufacture of paints, paper, plastics, etc.
Frequently Asked Questions – FAQs
What is the calcium carbonate used for?
It is used as an antacid is calcium carbonate to alleviate heartburn, acid indigestion, and stomach upset.
Name the fruit which is rich in calcium?
Apricot, gooseberry, figs and raisins are fruits rich in calcium.
Is Limestone a calcium carbonate?
It is a sedimentary carbonate rock that often consists of skeletal parts of marine organisms such as coral, foraminiferous, and molluscs.
How do you identify carbonate salt?
Strong acids, such as hydrochloric acid, can be added to assess whether a mineral or rock contains calcium carbonate. If the sample contains calcium carbonate, carbon dioxide will be fizzed and produced.
What is the formula for calcium carbonate?
The formula for calcium carbonate is CaCO3.
Learn more about Calcium Oxide, Calcium Sulphate and more with BYJU’S by downloading our app.


Calcium carbonate is one of the most popular chemicals. all equation are easily understood thanks for sharing this information.
Thanks for sharing basic useful information on calcite.