Preparation of a Colloidal Solution of Starch in Water and Egg Albumin/Milk in Water
- Solution: A solution is a combination of two or more substances that is homogenous in nature. Solid solutions, such as alloys, liquid solutions, such as lemonade, and gaseous solutions, such as air, are examples of possible solutions. Solute and solvent combine to form a solution.
- Solute: The solute is the part of the solution that is dissolved in the solvent.
- Solvent: Solvent is the component in a solution that dissolves the other components in it.
Aim:
To prepare a colloidal solution of starch in water and a colloidal solution of egg albumin in water and distinguish between them based on transparency, filtration criterion, and stability.
Theory:
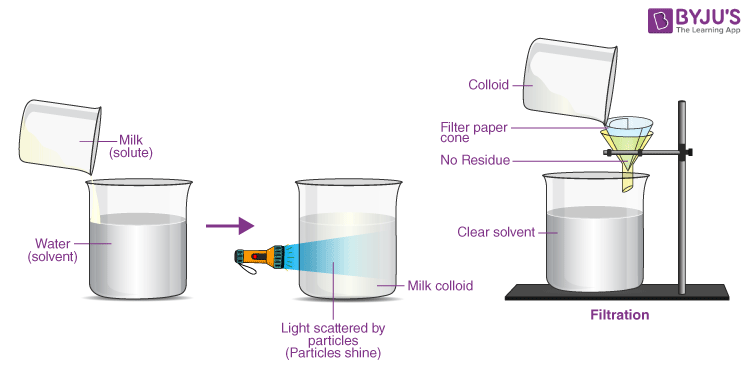
Colloidal solution appears to be homogeneous, the particles can scatter a ray of light, they do not settle down when left undisturbed, the solution is stable, and the particles are invisible to the naked eye. Filtering the particles is impossible. The diameter of the particles ranges from 10-7 cm to 10-4 cm.
Properties of Colloid:
- Although the solution is heterogeneous, it appears to be homogeneous.
- Solute particles range in size from 1 nm to 1000 nm. (10-9 – 10-6 m)
- The elements scatter light and produce the Tyndall effect.
- The only way to separate the particles is to use centrifugation.
- The solution is constant.
- The solution has a translucent appearance.
Materials Required:
Beakers, Test tubes, Starch, Egg albumin/milk, Glass Rod, Water.
Procedure:
For the colloidal solution of starch in water:
Step 1: In a beaker, mix 1% dry corn starch with 3 mL distilled water.
Step 2: Add 97 mL of boiling distilled water and mix thoroughly.
Step 3: After heating for 2 minutes, it is cooled and kept in test tube A.
For the colloidal solution of egg albumin/milk
Step 1: In a beaker, mix 1 gram of egg albumin/milk (the colourless fluid found around the yellow yolk) and 5 mL distilled water.
Step 2: Slowly pour in 5 mL of distilled water while stirring constantly.
Step 3: After mixing, add a few drops of acid (dil.HCl/dil.H2SO4 or acetic acid).
Step 4: The clear albumin and water solution became turbid and was stored in a test tube labelled B.
Observation Table:
|
Property |
Experimental Procedure |
Observation |
Inference |
|---|---|---|---|
|
Transparency |
Each test tube has a small strip of cellophane paper glued on it, and the coloured paper of each test tube can be seen from the other side. |
From the other side of test tubes A and B, the coloured spot appears dim. |
A colloid is translucent in nature. |
|
Filtration Criterion |
Filtrate the contents of test tubes labelled A and B. |
The resulting filtrate is translucent. In both situations, there was no residue on the filter paper. |
Filtration cannot separate the components that cause colloid. |
|
Stability |
Allow 20-25 minutes for the test tubes to rest without being disturbed. |
No change has been observed in test tubes A and B. |
When left undisturbed, colloids are stable, and solute particles do not settle down. |
Results and Discussion:
Colloidal solutions are translucent, and the particles in them can pass through filter paper to produce a translucent filtrate. There are no residue particles left on the filter paper.
Precautions to be taken during the experiment:
(i) Handle the materials and solutions with care.
(ii) While filtering a solution, pour the contents into the funnel using a glass rod.
(iii) Do not disturb the sample during the stability test.
Viva Voce
1. What is the Tyndall effect?
The Tyndall effect is referred to the scattering of a light beam.
2. Name the solute and solvent in the sugar solution.
The solute is sugar and the Solvent is water.
3. How can you separate colloidal particles from its solution?
By centrifugation.
4. Name three types of colloids.
Aerosol, foam, emulsion.
5. What will you name the heterogeneous solution whose particle size is between 10-7 cm to 10-4 cm?
It is a colloidal solution.
Comments