Table of Contents
What are Carbohydrates?Glycosaminoglycans DefinitionGlycosaminoglycans StructureGlycosaminoglycans FunctionApplications of GlycosaminoglycansGlycosaminoglycans Health Effects
Glycogen and Starch are composed of glucose units. Out of that, starch acts as storage form in plants, insoluble in water and digested with amylases. Glycogen is the storage form in animals. Cellulose is the main structural components in plants, and is indigestible by humans. Other polysaccharides like xanthan gum are present in the capsule of a bacterium.
They consist of protein cores which are made in the endoplasmic reticulum and post-translationally modified by the Golgi body. Glycosaminoglycan disaccharides are added to protein cores and form proteoglycans.
They are essential to life and important components of connective tissues. GAG chains are covalently bonded to other proteins like chemokines, cytokines, morphogens, growth factors, enzymes and adhesion molecules and forming proteoglycans.
What are Carbohydrates?
Carbohydrates are bio polymer composed of monomer units called monosaccharides. On the basis of number of monosaccharide units, carbohydrates can be classified into various types.
- Monosaccharides: Monomer units cannot further hydrolyse.
- Oligosaccharide: Hydrolysis of oligosaccharides gives 2-10 units of monosaccharides.
- Polysaccharides: They are composed of 10-100 or more than those units of monosaccharides.
Polysaccharides are complex carbohydrates which consist of multiple monosaccharides with other structures. They are also called as glycans as the monosaccharide units are bonded with glycosidic linkages. Polysaccharides are large and branched molecules which are often insoluble in water, amorphous in nature and non-sweet carbohydrates. Starch, cellulose, glycogen and chitin are the best examples of polysaccharides.
Polysaccharides can be classified into two types.
- Homopolysaccharides
- Heteropolysaccharides
Homopolysaccharides are composed of the same type of monosaccharide units while heteropolysaccharides have different types of monosaccharides. The general formula of polysaccharides is Cn(H2O)n-1 where n is a number between 200 and 2500.
Glycosaminoglycans Definition
Glycosaminoglycans are long unbranched polysaccharides which are composed of repeating disaccharide units and are also called GAGs or mucopolysaccharides due to their viscous and lubricating properties, just like in mucous secretions.
- They are found in collagen and elastin and water sticks to GAGs which allows resistance to pressure.
- Hence in the aqueous solution of GAGs, during compression, water is squeezed out and the GAGs are forced to occupy a smaller volume. As the compression is removed, they regain their original hydrated volume due to the repulsion arising from their negative charges.
- The repeating units of glycosaminoglycans consist of disaccharides units; one monosaccharide units is a hexose carbon sugar ring or a hexuronic acid which further bonded with a hexosamine; a six-carbon sugar containing nitrogen and one (or both) of the monosaccharide units contains at least one negatively charged sulphate or carboxylate group.
- Glycosaminoglycans are involved in a variety of extracellular and intracellular functions. For example; Heparin is a glycosaminoglycan which contains the highest net negative charge of the disaccharides and acts as a natural anticoagulant substance.
- It can bond strongly to antithrombin III (a protein involved in terminating the clotting process) and inhibits blood clotting. Another example is Hyaluronate molecules consist of around 25,000 disaccharide units with molecular weights of up to 107.
- They are important components of the vitreous humor in the eye and of synovial fluid which is a lubricant fluid of joints in the body. The keratan sulphate and chondroitins are also examples of glycosaminoglycans found in connective tissue like cartilage and tendons.
Similarly, dermatan sulphate is a component of the extracellular matrix of the skin. Out of these glycosaminoglycans, hyaluronic acid is not linked to a protein core while other three chondroitin sulphate, dermatan sulphate and Heparan sulphate are connected to proteoglycan through a serine residue.
Glycosaminoglycans Structure
Generally, glycosaminoglycans are linear, negatively charged polysaccharides which can sulfate or non- sulfate and have molecular weights of roughly 10-100 kilodalton.
On the basis of their structure units and linkage between disaccharide units; they can be classified into two types.
- Non-sulfated GAGs: For example, hyaluronic acid (HA)
- Sulphated GAGs: For example; chondroitin sulphate (CS), dermatan sulphate (DS), keratan sulphate (KS), heparin and heparin sulphate (HS).
There are disaccharides repeating regions in glycosaminoglycans chains which are composed of uronic acid-like D-glucoronic acid or L-iduronic acid and amino sugar like D-galactosamine or D-glucosamine. All these glycosaminoglycans are differing in the type of hexosamine, hexose or hexuronic acid unit as well as the geometry of the glycosidic linkage between these units.
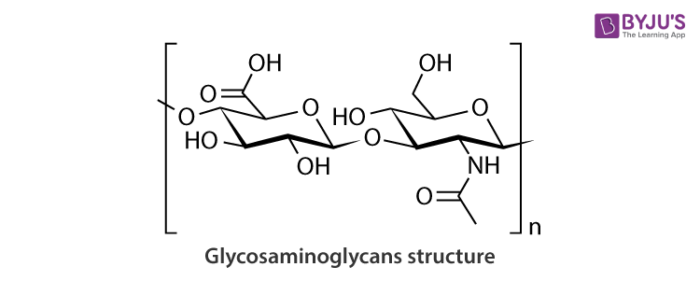
For example, dermatan sulphate and Chondroitin sulphate contain galactosamine and also known as glycosaminoglycans. Other glycosaminoglycans such as heparin sulphate and heparin contains glycosaminoglycan unit and are known as glycosaminoglycans.
- Chondroitin sulphate consists of beta-D-glucuronate linked to the third carbon of N-acetylgalactosamine-4-sulfate and heparin is a complex mixture of linear polysaccharides which have anticoagulant properties and vary in the degree of sulfation of the saccharide units.
- The amino sugar in glycosaminoglycans may be sulphated on C4 or C6 or on the non-acetylated nitrogen.
- The sugar backbone of GAGs can be sulphated at various positions; hence a simple octasaccharide can have over 1000 000 different sulphation sequences.
- In the repeating unit of each glycosaminoglycan species, there are one to two or 2 to 3 possible sulphation positions on the uronic acid and the amino sugar, respectively.
- Since these positions are not always sulphated, therefore 16 to 48 different disaccharide units can exist depending on the combination of sulfation positions.
Glycosaminoglycans Function
- Glycosaminoglycans (GAGs) participate in many biological processes through the regulation of their various protein partners called proteoglycan.
- The large structural diversity of GAGs makes them approachable for biochemical, structural biology and molecular modelling and made them useful in the discovery of new drugs.
- The conformational flexibility and underlying sulfation patterns of GAGs are responsible for the complexity of GAG-protein interaction.
- Four major negatively charged molecules made in animal cells are Glycosaminoglycans (GAGs), phospholipids and nucleic acids i.e., Ribonucleic acid and deoxyribonucleic acid.
- The negatively charged glycosaminoglycans cover the animal cell surfaces and interact with hundreds of extracellular signalling molecules.
- Due to their structural complexity, they have been claimed to be the most information-dense biopolymers found in nature. These biomolecules are related to transgenic and knockout animal data from the past decade which provides compelling evidence.
Applications of Glycosaminoglycans
Currently, many new projects are going on based on different applications of glycosaminoglycans.
Some of them are as follows.
- In the regulation of FGF/FGFR signalling.
- GAG-Based Activators and Inhibitors in FGF/FGFR Signaling.
- GAG Basis of Joint Specificity in Rheumatoid Arthritis.
- Serum GAGs and Proteoglycans as Biomarkers for lung Cancer.
Hyaluronic acid shows many important functions in signalling activity during embryonic morphogenesis, wound healing and pulmonary and vascular diseases. It also acts as lubrication of synovial joints and helps in joint movement as well as a space filler, wetting agent and flow barrier within the synovium. It also influences cancer progression and protector of cartilage surfaces.
Glycosaminoglycans Health Effects
- CD44(cell-surface glycoprotein), which is expressed on the surface of virtually all stem cells including cancer stem cells acts as the main receptor for Hyaluronic acid. The interaction of hyaluronic acid can mediate leukocyte rolling and extravasation in some tissues. Other glycosaminoglycans, heparin was first discovered in 1917 and mainly used for anti-coagulation.
- The anti-coagulation property of sulfated GAGs is because of its capacity to prolong the process of blood clotting which is due to the potentiating interaction of GAGs with the natural inhibitor of thrombin, antithrombin III (AT-III), with only about one-third of all heparin chains possessing the structures required for AT binding.
- Hence heparin is mainly used as an anticoagulant for the treatment of thrombophlebitis, embolism and thrombosis. Heparin was isolated from canine liver cells and derived from bovine lung or porcine intestinal mucosa.
- Glycosaminoglycans play a vital role in cell signalling and development, angiogenesis, anti-coagulation, tumour progression, axonal growth and metastasis. They also involve in cell proliferation as they act as co-receptors for growth factors of the fibroblast growth factor family. The members of the fibroblast growth factor family need to interact with both a heparin ⁄ HS chain and realize their full signalling potential by their high-affinity receptor.
- Sulphated glycosaminoglycans are a common constituent in many different types of amyloid which play an important role in the pathology of amyloid diseases like amyloid A-amyloidosis, prion diseases, type-2 diabetes and Parkinson’s disease. During these diseases, there will be deposition in tissues of fibrillar aggregates of polypeptides. Glycosaminoglycans like Heparan sulphate can bind with amyloidogenic peptides in vitro and in vivo to promote the fibril formation and enhance the disease condition.
- Diseases like inflammatory bowel disease, and microbial infections and rheumatoid arthritis are also associated with inflammatory responses and many proteins play a role in the inflammation cascade which leads to the activation of leukocytes and endothelial cells and ultimately to the extravasation of leukocytes and leukocyte migration into the inflamed tissue. Glycosaminoglycans like heparin act as an adhesion ligand in leukocyte extravasation and carriers of chemokines and growth factors.
