What is Methylene blue (C16H18ClN3S)?
Methylene blue is an organic chloride salt with a formula C16H18ClN3S. It is also called Methylthioninium chloride or Swiss Blue.
It is a thiazine dye with antioxidant, cardioprotective properties, antimalarial, and an antidepressant. It is soluble in glycerol, water, chloroform, glacial acetic acid, and ethanol. It is slightly soluble in pyridine and insoluble in ethyl ether, oleic acid, and Xylene.
Table of Contents
- Properties of Methylene blue-C16H18ClN3S
- structure of Methylene blue-C16H18ClN3S
- Uses of Methylene blue (C16H18ClN3S)
- Effects on Health
- Frequently Asked Questions
It is a synthetic basic dye. When its administration route is intravenously and in low doses, it converts methemoglobin to haemoglobin. It functions as a histological dye, tracer, acid-base indicator, neuroprotective agent, fluorochrome, etc.
This compound was first synthesized by Heinrich Caro in the year 1876. It is obtained as a dark green crystalline powder or crystal with a bronze-like lustre. When dissolved in an alcohol solution or water it gives a deep blue colour. It is widely used in treating methemoglobin levels greater than 30%.
Properties of Methylene blue – C16H18ClN3S
| Methylene blue | C16H18ClN3S |
| Molecular Weight of Methylene blue | 319.85 g/mol |
| Trade names | Urelene blue, Proveblue, Provable |
| Melting Point of Methylene blue | 100 to 110 °C |
| Routes of administration | Mouth and IV |
Structure of Methylene blue (C16H18ClN3S)
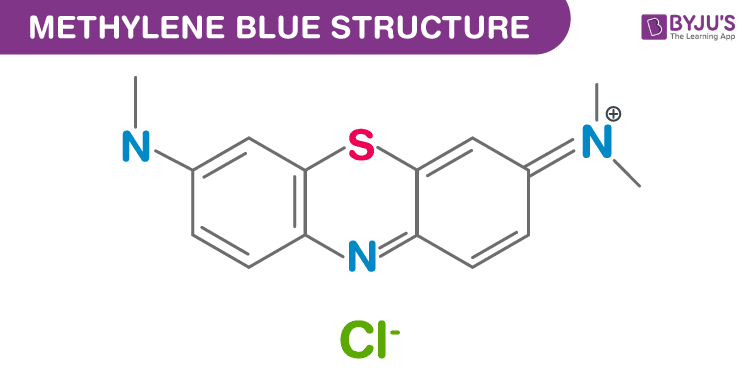
Structure of Methylene blue – (C16H18ClN3S)
Uses of Methylene blue (C16H18ClN3S)
- Methylene blue is used as an indicator
- It is used as a bacteriologic stain.
- Used to lower levels of Methemoglobin.
- It is combined with light and used in the treatment of resistant plaque psoriasis.
- It is used to identify dysplasia.
Effects on Health
Common side effects include headache, vomiting, high blood pressure, confusion, allergic reactions, shortness of breath, red blood cell breakdown, serotonin syndrome. Using this turns the urine, stool, and sweat, blue to green in colour. It converts the ferric iron present in the haemoglobin to ferrous iron.
Frequently Asked Questions
What is methylene blue used for?
Methemoglobinemia can occur when a person is exposed to certain drugs or chemicals like nitrites. Methylene blue injection is used to treat methemoglobinemia. Methylene blue works by transforming methemoglobin into a more effective form of haemoglobin to help hold oxygen all over the body.
Is methylene blue radioactive?
The widely used method isosulphan blue / patent blue V paired with colloid tracer radioactive. Methylene blue (MB) is a less costly replacement dye, which is readily available. The dye and radioisotope were injected into the subdermal plane in the subareolar region.
Is methylene blue a carcinogen?
Methylene blue trihydrate was not properly tested for carcinogenicity, given its use as a therapeutic. In addition, there is a lack of epidemiological research relating to methylene blue carcinogenicity.
What are the side effects of methylene blue?
Large intravenous doses of methylene blue injection induce nausea, abdominal and precordial discomfort, dizziness, headache, profuse sweating, mental confusion, and methemoglobin formation.
What is the purpose of methylene blue dye?
Methylene blue is a widely used stain which lets us see brilliant colour in microscopic life. Until putting the slide under the microscope, biologists often apply a drop or two of methylene blue to bacteria on a glass slide. The blue colour that stains the bacteria assists biologists in seeing their forms.
Learn more about the physical and chemical properties of Methylene blue (C16H18ClN3S) from the experts at BYJU’S.

Comments