What is a Diazonium Salt?
Diazonium salts are a group of organic compounds which share a common functional group R – N2+ X– where R denotes some organic residue (such as an alkyl or aryl group) and X is an organic or inorganic anion (a halogen, for example). Diazonium salts in which R is an aryl group are significant intermediates in the organic synthesis of azo dyes. They are named by adding a suffix diazonium to the parent hydrocarbon from which they are formed, followed by the name of the anion such as chloride or hydrogen sulphate. The ion N2+ is commonly referred to as a diazonium group. For example, C6H5N2+Cl– is known as benzene-diazonium chloride. Diazonium salts are colourless crystalline solids and are readily soluble in water. They decompose easily in a dry state.
Chemical Reactions of Diazonium Salts
Diazonium salts undergo two types of reactions:
- Reactions which involve the replacement of nitrogen
- Reactions which involve retention of the diazo group.
This article deals with the reactions which involve the replacement of nitrogen. Diazonium group is a good leaving group. They are substituted by other groups such as Cl–, Br–, CN–, and OH– which easily displace nitrogen from the aromatic ring. This nitrogen escapes from the reaction mixture in the form of a gas.
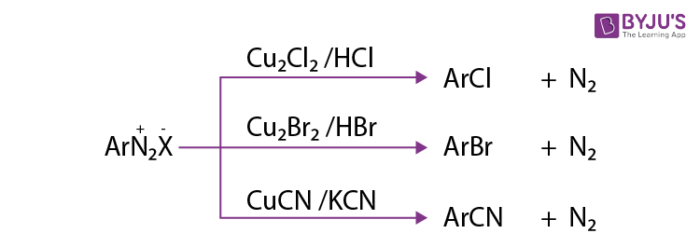
1. Replacement by halide or cyanide ion: The nucleophiles such as Cl–, Br–, CN– can be easily introduced into the benzene ring in the presence of Cu (I) ion. This reaction is commonly known as the Sandmeyer reaction.
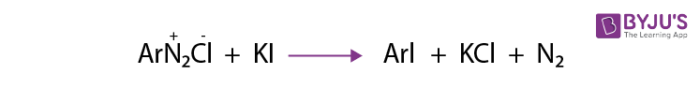
 2. Replacement by iodide ion: It is not easy to introduce iodine in the benzene ring directly. But when diazonium salt solution is treated with potassium iodide, iodobenzene is formed.
2. Replacement by iodide ion: It is not easy to introduce iodine in the benzene ring directly. But when diazonium salt solution is treated with potassium iodide, iodobenzene is formed.
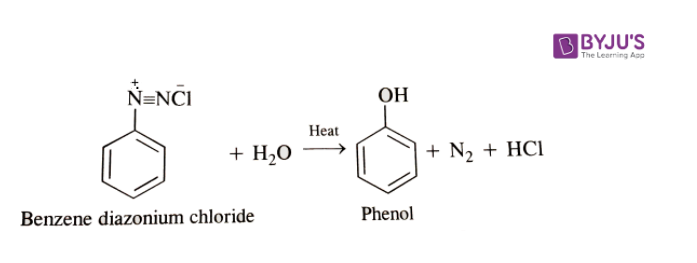
 3. Replacement by hydroxyl group: If the temperature of the diazonium salt solution is increased to 283 K, the salt gets hydrolyzed to phenol.
3. Replacement by hydroxyl group: If the temperature of the diazonium salt solution is increased to 283 K, the salt gets hydrolyzed to phenol.
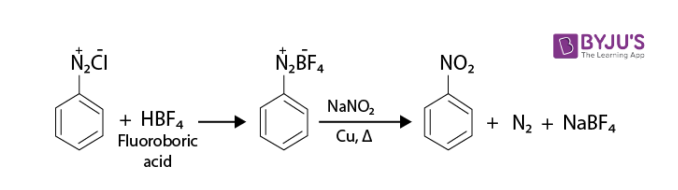
4. Replacement by –NO2 group: When we heat diazonium fluoroborate with an aqueous sodium nitrite solution in the presence of copper, the diazonium group is replaced by –NO2.
This article covers the reaction of diazonium salts along with its basic properties. For any further information on this topic install BYJU’S – The Learning App.

Comments