“Carboxylic acids” is one of the important chapters in organic chemistry for JEE. Questions related to its preparation, physical and chemical properties are asked almost every year in JEE.
Important Questions For Carboxylic Acids
Some FAQs related to carboxylic acids are underlined below:
Question: What are carboxylic acids?
Answer: Carboxylic acid is an organic compound containing a carboxyl group (COOH) attached to an alkyl or aryl group. The general formula of a carboxylic acid is CnH2n+1COOH. In carboxylic acids, three of the four bonds of the carboxyl carbon atom are with two oxygen atoms.
Question: Explain the physical properties of carboxylic acids?
Answer: Carboxylic acids have high boiling points as compared to aldehydes and ketones of comparable masses because of intermolecular hydrogen bonding. The simple aliphatic carboxylic acids are soluble in water due to the formation of hydrogen bonds in water. As the number of carbon atoms in carboxylic acids increases their solubility decreases due to an increase in the length of the hydrophobic group attached to the carboxyl group.
Question: Explain the chemical properties of carboxylic acids?
Answer: Carboxylic acids are highly acidic in nature. Their acidity is higher than that of phenols and alcohols but lesser than that of mineral acids. They generally undergo four types of reactions:
- Reactions involving cleavage of O-H bond, for example, esterification of carboxylic acids.

- Reactions involving cleavage of C-OH bonds, for example, reaction with ammonia.
CH3COOH + NH3 → CH3CONH2
- Reactions involving degradation of –COOH group, for example, decarboxylation.
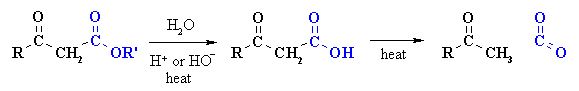
- Reactions involving substitution in the hydrocarbon part, for example, Hell-Volhard-Zelinsky reaction (halogenation).

Questions: Which reference books can one follow for carboxylic acids?
Answer: While covering carboxylic acids for JEE, one must go through the NCERT class 12th chemistry textbook part-2. A brisk walk through this chapter from NCERT not only gives you a sound idea of the various topics you need to cover according to the JEE syllabus but also develops the fundamentals regarding the chapter Apart from this you can follow a reference book to understand the mechanism of the reactions not given in the NCERT textbook. Some authentic books for understanding the mechanism of organic reactions include books from authors like Solomons & Fryhle, Morrison & Boyd. Once you have developed concepts on this topic, you can practice questions on this topic from the books of authors like M. S.Chauhan.
For further details related to carboxylic acids for JEE chemistry, get in touch with our mentors here at BYJU’S.
Comments