Hoffmann bromamide reaction is one of the most important reactions in organic chemistry, included in JEE syllabus. It is one of the major reactions for the synthesis of primary amines. Some FAQs related to Hoffmann bromamide reactions are given below:
Important Hoffmann Bromamide Questions
What Is Hoffmann bromamide Reaction?
When an amide is treated with bromine in an aqueous or ethanolic solution of sodium hydroxide, degradation of amide takes place leading to the formation of primary amine. This reaction involves the degradation of amide and is popularly known as Hoffmann bromamide degradation reaction. Thus, the primary amine contains one carbon less than the number of carbon atoms in that amide.
RCONH2 +Br2 + 4NaOH → R-NH2 + Na2CO3 + 2NaBr + 2H2O
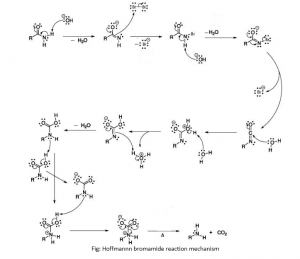
What Is The General Mechanism Of Hoffmann Bromamide Reaction?
The general mechanism of the Hoffmann bromamide reaction includes the following steps:
- A strong base (usually an alkali) attacks the amide, this leads to deprotonation generating an anion.
- This anion reacts with bromine to form N-bromoamide. This reaction can be seen as an α-substitution reaction.
- Deprotonation of the bromoamide molecule to generate a bromoamide anion.
- The generated bromoamide anion undergoes rearrangement such that the R group attached to the carbonyl carbon migrates to nitrogen. The bromide ion leaves the compound simultaneously, leading to the formation of isocyanate.
- Water molecules are added to the isocyanate to form carbamic acid. This reaction is an example of nucleophilic addition.
- Finally, the carbamic acid loses carbon dioxide, CO2 leading to the formation of primary amine.
Which Reference Books Can One Follow For Understanding Hoffmann Bromamide Reaction?
For understanding the Hoffmann bromamide degradation reaction, one must go through the NCERT class 12th chemistry textbook part 2. A brisk walk through NCERT will give you a sound idea of this reaction. Apart from this, you can follow a reference book to understand the detailed mechanism of this reaction. Some authentic books for understanding the detailed mechanism of Hoffmann bromamide reaction include organic chemistry books from authors like Solomons & Fryhle, Morrison & Boyd. Once you have understood the reaction, you can practice questions from the books of authors like M. S. Chauhan, etc.
Stay tuned with BYJU’S for further details related to Hoffmann bromamide reaction for JEE chemistry, Important books for JEE Chemistry, Frequently Asked Questions in Inorganic chemistry, JEE Preparation, Important JEE Chemistry topics etc.
Comments