Table of Content:
| Battery | Electrode | Positive and Negative terminals | Cathode or Negative Terminal | Anode or Positive Terminal |
Battery
A battery is an electrochemical device that stores energy in the form of chemical energy and gives out electrical energy. The battery has a positive terminal, and a negative terminal and the electrolyte is the chemical solution which surrounds the anode and the cathode. The current starts flowing in the battery when the positive terminal and negative terminal are connected.
A battery terminal is an electrical contact that can be used to connect a single or multiple-celled battery. The battery terminals come in a variety of sizes, designs and features. However, the two main configurations of a battery terminal are – positive on the left and negative on the right, or negative on the left and positive on the right. For example, the positive and negative terminals of a dry battery have a projection at one end (indicating positive terminal) and a flat base on the other end that indicates negative terminal.
These batteries are of different types. An example of a primary battery is the dry cell. In a dry cell, a zinc container is used as the anode, and a carbon rod is used as the cathode. The space around the cathode is surrounded by manganese dioxide and the cathode. The space between the container and the rod is filled with a moist paste of ammonium chloride and zinc chloride.
Electrode
The electrode, usually metal, is the electrical conductor used as a positive or negative terminal of an electrically conducting medium. The electrodes conduct the current into the medium and out of the medium. The medium is either solid, gas, vacuum or an electrolytic solution, as in a storage battery.
Positive and Negative terminals
The electrode from which the electrons emerge is the negative terminal. It is also called the cathode. The electrode that receives electrons is the positive terminal. It is called the anode.
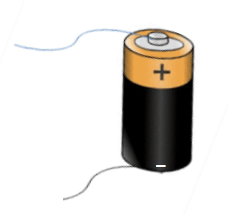
Cathode or Negative Terminal
- The cathode is the negatively charged electrode
- It attracts the positive charge or cations
- The cathode is the source of the electrons
- The cathode generates the electrons, which do the actual movement in a circuit and produces current. Therefore, we can say that the cathode produces the charge or that the current moves from the cathode to the anode. Though the conventional direction of current is defined by the way, a positive charge would move.
Anode or Positive Terminal
- The anode is the positively charged electrode.
- It attracts electrons or anions
It helped me a lot!