Table of Contents
What is Chloroform?
Chloroform is an organic chemical compound initially employed as an ideal anaesthetic. It was first prepared in 1831. The chemical formula is CHCl3. It is a colourless, sweet-smelling dense liquid produced on a large scale. Chloroform may be released into the air as a result of its formation in the chlorination of drinking water, wastewater and swimming pools. Chloroform is an industrial chemical that can act as a lacrimator. It is non-flammable, which makes it safer to handle than ethanol.
Other names – Trichloromethane, Formyl trichloride, Trichloroform
| CHCl3 | Chloroform |
| Density | 1.49 g/cm³ |
| Molecular Weight/ Molar Mass | 119.38 g/mol |
| Boiling Point | 61.2 °C |
| Melting Point | -63.5 °C |
| Chemical Formula | CHCl3 |
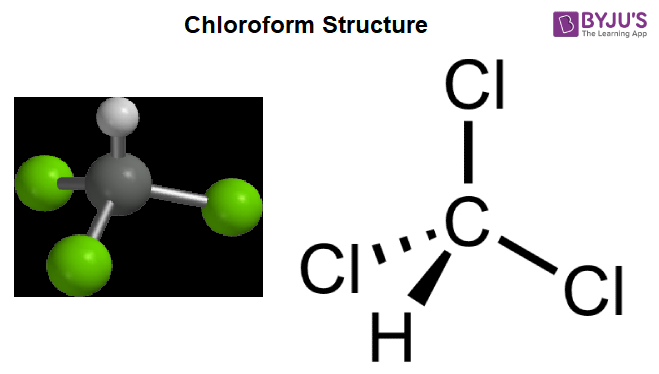
Chloroform Structure – CHCl3
Physical Properties of Chloroform – CHCl3
| Odour | Ether-like odour |
| Appearance | Clear liquid |
| Covalently-Bonded Unit | 1 |
| Complexity | 8 |
| Heavy Atom Count | 4 |
| Solubility | Insoluble in water |
Chemical Properties of Chloroform – CHCl3
-
-
-
- Chloroform reacts with base sodium hydroxide forms sodium formate, sodium hydroxide and water. The chemical equation is given below.
-
-
CHCl3 + 4NaOH → HCOONa + 3NaCl + 2H2O
-
-
-
- Chloroform on oxidation forms phosgene and hydrochloric acid. The chemical equation is given below.
-
-
Uses of Chloroform – CHCl3
-
-
- Used as an anaesthetic and used in dentistry during root canal procedures.
- The spectrum of pure chloroform is used as the reference or background, and pure cholesterol powder or cholesterol extract from milk products is dissolved in chloroform and used for FTIR analysis.
- Chloroform was utilized in the past as an extraction dissolvable for fats, greases, oils, and different items; as a laundry spot.
- Used as an indirect food additive in food packaging materials for adhesive components and as a component of food contact materials.
-
Frequently Asked Questions
What are the uses of chloroform?
Chloroform is a volatile and colourless liquid which is a trichloromethane derivative with a smell similar to ether. Formerly used during surgery as an inhaled anaesthetic, the main application of chloroform today is in agriculture, where it is used as a solvent, and also particularly in the manufacture of the refrigerant freon.
How does chloroform affect the human body?
Local symptoms following chloroform inhalation include shortness of breath, and nose and throat irritation. Acute inhalation can lead to systemic symptoms such as agitation, nausea and vomiting accompanied by ataxia, dizziness, and drowsiness.
Comment on the physical appearance of chloroform
Chloroform is a transparent liquid with a slightly sweet taste and an ether-like odour. It is a naturally occurring compound but much of the environmental chloroform is man-made.
How does chloroform render people unconscious?
Chloroform effectively makes a human unconscious by disrupting the functions of intercellular protein synthesis. Doctors will use a cloth mask with a constant drip of chloroform (or ether) onto it, placed over the nose and mouth with a wire frame, to knock someone out with chloroform.

Thankyou very much it’s more helpful