What is Dimethyl sulfoxide?
Dimethyl sulfoxide also known as DMSO is a colourless highly polar organic liquid with the chemical formula C2H6OS that was discovered in 1866.
DMSO was first synthesized by Russian scientist Alexander Saytzeff in Kazan in central Russia. It has a high dielectric constant and therefore exhibits exceptional solvent properties for both organic and inorganic chemicals. It’s better stored in glass containers and protects from light and moisture because it is highly hygroscopic.
Other name – methylsulfinylmethane, methylthiomethane
| C2H6OS | Dimethyl sulfoxide |
| Density | 1.1 g/cm³ |
| Molecular Weight/ Molar Mass | 78.13 g/mol |
| Boiling Point | 189 °C |
| Melting Point | 19 °C |
| Chemical Formula | (CH3)2SO |
Dimethyl sulfoxide Structure
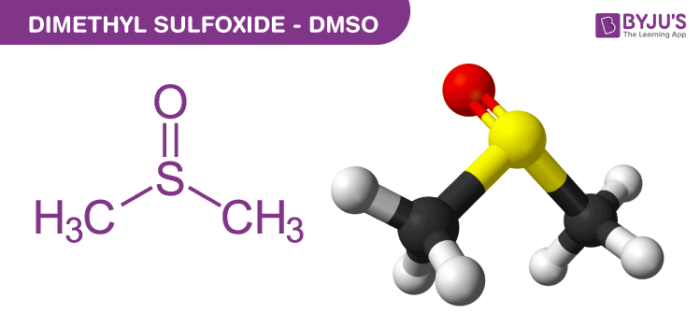
Physical Properties of Dimethyl sulfoxide – C2H6OS
| Odour | Cabbage-like |
| Appearance | Colorless liquid |
| Complexity | 29 |
| Specific Gravity | 1.101 at 20oC |
| Hydrogen Bond Acceptor | 2 |
| Solubility | Miscible in organic solvents as well as water. |
Chemical Properties of Dimethyl sulfoxide – C2H6OS
-
- Dimethyl sulfoxide is readily reacts with hydrogen peroxide forming thioglycolic acid and water. The chemical reaction is as below.
C2H6OS + 2H2O2 → C2H4O2S + 3H2O
Uses of Dimethyl sulfoxide – C2H6OS
- Used as a non-medicinal Industrial solvent for resins, fungicides, dyes, pigments.
- Used as a reactant for chemical synthesis Antifreeze Hydraulic fluid, Paint remover, Medicinal Anti-inflammatory agent.
- It was reported that DMSO penetrates through the skin and produces analgesia, decreases pain, and promotes tissue healing.
- DMSO is an abundant relatively cheap byproduct of the wood pulp industry with a primary use as an industrial solvent.
Frequently Asked Questions
What is dimethyl sulfoxide used for?
DMSO is used topically to relieve discomfort and to improve wound healing, burns, and muscle and skeletal injuries. Topically, DMSO is also used to treat painful conditions such as headache, swelling, osteoarthritis, rheumatoid arthritis and extreme facial pain called tic douloureux.
Is dimethyl sulfoxide toxic?
DMSO is a non-toxic solvent, with a higher than ethanol median lethal dose. Due to concerns about its health in particular, its potential to damage the eye, early clinical trials with DMSO were stopped
Why is DMSO toxic to cells?
DMSO is considered a solvent poor in toxicity. However, some DMSO concentrations may alter the activation of cells in culture. Cultures of cells derived from rats treated with DMSO increased lysosomal enzyme activity by 25 per cent
What is DMSO made from?
DMSO, or dimethyl sulfoxide, is a paper-making byproduct. It originates from a substance found in wood. Since the mid-1800s, DMSO has been used as an industrial solvent. DMSO has been approved by the FDA as a prescription drug for treating symptoms of painful bladder syndrome.
Is DMSO denser than water?
Such compounds are denser than water, and so occur later when combined with water in the centre. Not that certain organic compounds like dimethylformamide (DMF) and dimethylsulfoxide (DMSO) are so polar that they’re miscible with water (which is also very polar), and don’t form layers.

Comments