Table of Contents
Preparation of Carboxylic Acid?
The compound in which the carbonyl group is attached to hydroxyl then it is called Carboxylic acid. There are various methods that can be used for the preparation of Carboxylic acids.
By using primary Alcohols and Aldehyde
We will notice that the primary alcohol gets oxidised to carboxylic acid when oxidising agents are added, such as potassium permanganate (KMnO4), in an acidic, alkaline or neutral medium. By using a mild oxidising agent, we can get carboxylic acids from aldehydes.

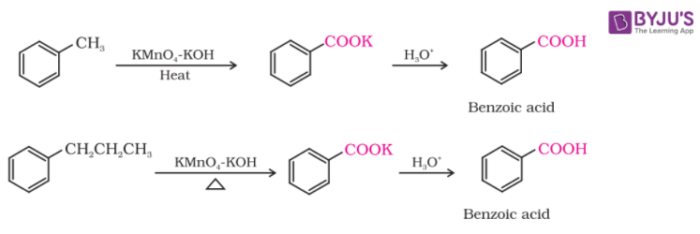
From Alkylbenzenes
We can get aromatic carboxylic acids after vigorous oxidation of alkylbenzenes with chromic acid. Irrespective of the length of the chain, the whole side chain is oxidised to a carbonyl group. The oxidation of primary and secondary alkyl groups can be done in this manner while the tertiary group is unaffected.
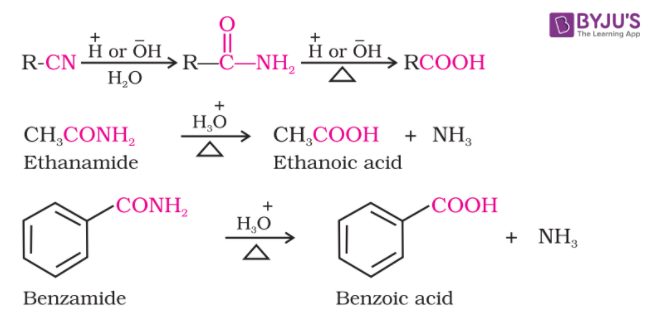
From Nitriles and Amides
Amides are prepared by the hydrolysis of nitriles and then converted to acids in the presence of catalysts (H+ or OH-1). In order to stop the reaction at the amide stage, mild reaction conditions are used.
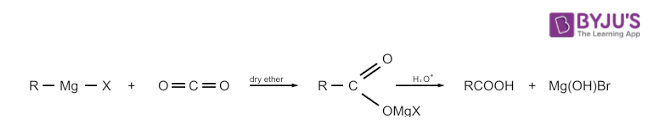
By using Grignard Reagents
When Grignard reagent reacts with carbon dioxide, it forms salts of carboxylic acids which after some time gives corresponding carboxylic acid after the acidification with any mineral acid. We can prepare Grignard reagents as well as nitriles from alkyl halides. These methods are very useful for the conversion of alkyl halides into corresponding carboxylic acids which has one carbon atom more than what is present in alkyl halides.
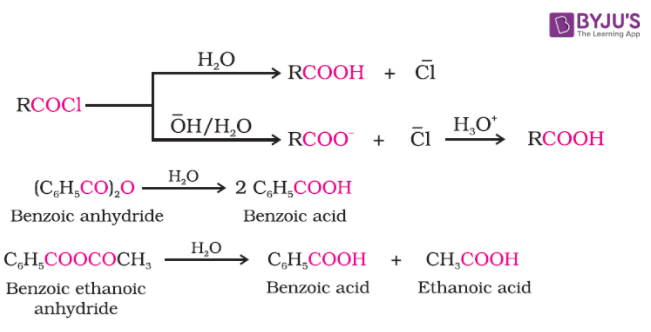
By Acyl Halides and Anhydrides
When acid chloride is hydrolysed with water then carboxylic acid is produced and it is more readily hydrolysed with an aqueous base and gives corresponding carboxylic ions which on further acidification gives the carboxylic acid.
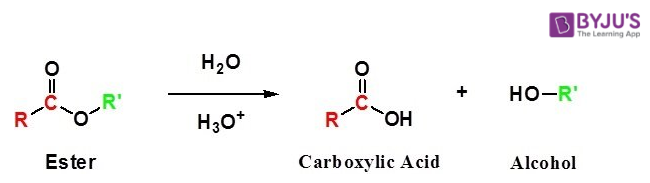
By using Esters
A carboxylic acid is produced after the acidic hydrolysis of esters and carboxylates are produced after the basic hydrolysis of an ester.

Comments