In order to understand the various chemical reactions that occur around us and the parameters governing these reactions, we need an understanding of the elements and compounds taking part in these reactions. The early developments in science revealed that matter is made up of atoms which in turn are composed of elementary particles known as electrons, protons and neutrons. In order to have a better understanding of the transformations of matter from one form to the other, scientists needed a defined structure for an atom and its interaction with other atoms. J.J. Thomson, an English scientist, proposed the famous Thomson atomic model in the year 1898 just after the discovery of electrons.
Plum Pudding Atomic Theory
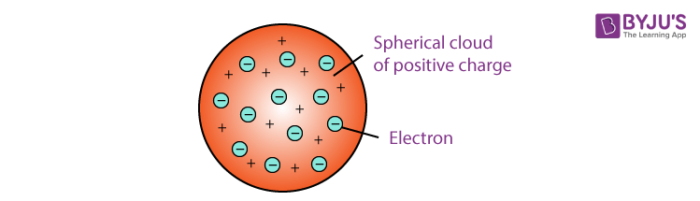
Thomson proposed that the shape of an atom resembles that of a sphere having a radius of the order of 10-10 m. The positively charged particles are uniformly distributed with electrons arranged in such a manner that the atom is electrostatically stable. Thomson’s atomic model was also called the plum pudding model or the watermelon model. The embedded electrons resembled the seed of a watermelon while the watermelon’s red mass represented the positive charge distribution. The plum pudding atomic theory assumed that the mass of an atom is uniformly distributed all over the atom.
Limitations of Thomson’s Atomic Model
Thomson’s atomic model explained the overall neutrality of an atom. Its assumption that the total mass of an atom is uniformly distributed all over the atom was inconsistent with some experimental results. Rutherford’s gold foil experiment of scattering α-particles revealed that most of the space in an atom is empty, contradicting Thomson’s atomic model. Thus, it was found that the plum pudding atomic model was insufficient to explain the structure of an atom.
The limitations of Thomson’s atomic model triggered further research for establishing the structure of an atom. The plum pudding atomic model lacked experimental evidence and hence Rutherford conducted many experiments in order to determine the structure of an atom.
Recommended Videos
JJ Thomson Atomic Model and Its Limitations

Log on to BYJU’S YouTube channel to learn more about Rutherford’s atomic model that came after Thomson’s atomic model failed to explain the structure of an atom.

THANKS A LOT !!!!!!!
Nice