What is Polyurethane?
Polyurethane, often abbreviated to PU or PUR, is an organic polymer that features many organic units linked via urethane molecules. Most polyurethanes do not melt upon heating and can, therefore, be classified as thermosetting polymers. However, it can be noted that some specific types of polyurethanes exhibit thermoplastic properties and can be melted and remoulded via the application of heat.
Traditional methods of preparing polyurethanes involve chemical reactions between polyols and di- or tri-isocyanates. Polyurethanes can be regarded as alternating copolymers since they feature two kinds of monomers that undergo polymerization one after the other. It can also be noted that both the polyols and the isocyanates that are used as monomers for the production of polyurethanes usually contain at least 2 functional groups in every molecule.
Preparation of Polyurethane
Polyurethanes, along with phenolics, epoxies, and unsaturated polyesters, can be classified as reaction polymers. They can be prepared via the chemical reactions between diisocyanates (that contain at least two isocyanate groups per molecule) and polyols (that contain at least two hydroxyl groups per molecule). These reactions usually require the presence of a catalyst or some ultraviolet light in order to overcome the activation energy barrier.
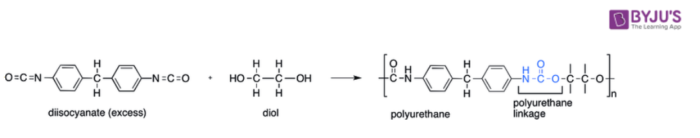
Polyols, by themselves, can also be considered polymers. For example, polyether polyols can be prepared by subjecting propylene oxide and ethylene oxide to copolymerization along with an appropriate polyol precursor. It can also be noted that the preparation of polyester polyols is quite similar to that of polyether polyols. However, for the production of polyurethanes, the chain length of the polyol and the functionality of the polyol must be controlled. These factors are known to contribute to the properties of the final polyurethane products. For example, the polyols whose molecular weights lie in the hundreds yield rigid polyurethanes whereas the polyols whose molecular weights lie in the thousands yield relatively flexible polyurethanes. Thus, the chain length of the polyol used can be considered a factor that contributes to the flexibility of the polyurethane product.
Properties of Polyurethane
The properties of polyurethanes are highly dependent on the manner in which they are produced. For example, if the polyol chain (which is used as a raw material for the preparation of the polyurethane) is long and flexible, the final product will be soft and elastic. On the other hand, if the extent of cross-linking is very high, the final polyurethane product will be tough and rigid. The cross-linked structure of polyurethanes generally consists of three-dimensional networks which attribute very high molecular weights to the polymer. This structure also accounts for the thermosetting nature of the polymer, since polyurethane typically does not soften or melt when exposed to heat.
Advantages of Polyurethanes
Polyurethanes have the ability to be made into foams (which is one of their most desirable properties). This is done by facilitating the production of a gas (usually carbon dioxide) during the urethane polymerization process. Another advantage of polyurethane is that high-density microcellular foams of the polymer can be produced without any blowing agents.
Applications of Polyurethane
Some important applications of polyurethane are listed below.
- The primary application of polyurethane is in the production of foams. These foams are used in a variety of materials such as upholstery fabrics, domestic furniture, and refrigerator sheets.
- Polyurethane is also used in some garments.
- Polyurethane mouldings are also used in columns and door frames. In fact, it is not uncommon for such mouldings to be employed in window headers and balusters.
- The low-density foams of polyurethane which exhibit flexibility are widely used in mattresses and other forms of bedding. They are also used in automobile seats and upholstery.
- Flexible polyurethane is also used in the manufacture of partially elastic straps and bands.
- The low-density elastomers of polyurethane are widely used in the footwear industry.
- Another notable application of polyurethane is in the manufacture of bathroom and kitchen sponges. It is also used in seat cushions and couches.
Frequently Asked Questions on Polyurethane
What is thermoplastic polyurethane?
Thermoplastic polyurethane, often abbreviated TPU, is a class of polyurethane which are made up of thermoplastic elastomers. Their structures usually feature linearly segmented block copolymers that are made up of soft and hard segments. The desirable properties of thermoplastic polyurethanes include elasticity, resistance to oil, resistance to grease, transparency, and resistance to abrasion. TPUs are widely used in power tools, medical devices, sporting goods, footwear, caster wheels, and inflatable rafts. Another notable application of thermoplastic polyurethane is in cable and wire jacketing.
What are the uses of polyurethane?
Flexible polyurethane foam is widely used as padding in a wide range of consumer and industrial items including bedding, underlay carpets, vehicle interiors, chairs, and packaging. This type of polyurethane foam can be produced in virtually any shape and firmness. It is known to be lightweight, comfortable, and durable. Rigid polyurethane foams are used in highly energy-efficient and flexible insulations. These foams are known to significantly reduce energy costs in residential and commercial properties.
How is polyurethane prepared?
Polyurethanes are prepared by reacting polyols (alcohols with more than two reactive hydroxyl groups in each molecule) with diisocyanates or polymeric isocyanates. Suitable catalysts and additives are used wherever necessary. Since a variety of diisocyanates and a wide range of polyols can be used for the production of polyurethane, a wide spectrum of polyurethane materials can be produced to meet the requirements for specific applications. It can also be noted that polyurethanes can exist in a variety of forms including rigid foams, flexible foams, speciality adhesives, chemical-resistant coatings, sealants, and elastomers.
To learn more about polyurethane and other commercially important polymers such as bakelite, register with BYJU’S and download the mobile application on your smartphone.

Comments