Benzene is a hydrocarbon with the chemical formula C6H6. It has 6 carbon atoms joined in a ring and has 1 hydrogen atom attached to each of the carbon atoms. By replacing one or more of the hydrogen atoms with some functional group, we get several benzene compounds. While naming the substituted benzene compounds, we prefix the name of the substituent to the word benzene. In this article, we will learn the IUPAC nomenclature of the substituted benzene compounds.
Table of Contents
- Mono-substituted Benzene Compounds
- Di-substituted Benzene Compounds
- Poly-substituted Benzene Compounds
- Recommended Videos
Mono-substituted Benzene Compounds
For the benzene compounds consisting of a single substituent, we simply prefix the name of the substituent to benzene. Some examples along with their common names are listed below.
-
Methylbenzene or Toluene

Toluene
-
Hydroxybenzene or Phenol

Phenol
-
Aminobenzene or Aniline

Aniline
-
Ethylbenzene
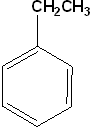
Ethyl benzene
Di-substituted Benzene Compounds
When there are two substituents present in the compound, we number each of the carbon atoms in such a manner that the substituents are attached to the lowest possible numbered carbon atom.
- 1,3-dinitrobenzene is the name of the following compound. Naming it as 1,5-dinitrobenzene is incorrect because two nitro groups must have the lowest numbers.
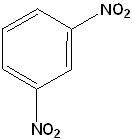
- 1,3-dichlorobenzene
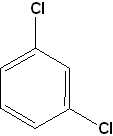
Moreover, when there is more than one substituent, we also name their positions as ortho- (o), meta- (m) and para- (p). They refer to the positions 1,2-;1,3- and 1,4- respectively. Thus, we can name the compound 1,3-dichlorobenzene as m-dichlorobenzene.
Poly-substituted Benzene Compounds
In the case of poly-substituted compounds, if there is a base compound present, we assign it as position 1. We then choose the next compound for numbering such that it gets the lowest number. However, if there are no special or base group present, we list them in alphabetical order, giving them the lowest numbers.
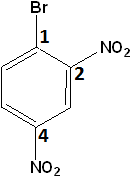
-
1-Bromo-2,4-dinitrobenzene
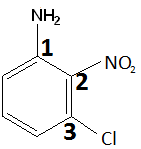
- For the following compound, aniline is the base compound. So, we name it as 3-chloro-2-nitroaniline.
Recommended Videos
Electrophilic Substitution Of Benzene

To learn more about benzene, its compounds and their nomenclature, download BYJU’S – The Learning App.

Comments