Introduction
Swern oxidation offers a way to oxidize alcohols without the usage of toxic metals (chromium, for example). Very mild conditions are required to carry out this process, which is another advantage.
Table of Contents
What is Swern Oxidation?
Swern Oxidation refers to the organic chemical reaction where oxalyl chloride, an organic base, and dimethyl sulfoxide are employed for the oxidation of primary and secondary alcohols to aldehydes or ketones, respectively. It is important to note that these aldehydes do not undergo further reaction to produce carboxylic acids.
A disadvantage of this reaction is the formation of dimethyl sulphide as a side product, which has an unpleasant smell which can become highly disagreeable even in low concentrations. The reaction is an organic redox reaction. The Swern oxidation reaction is named after its discoverer – the American chemist Daniel Swern. An example of the Swern reaction is given below.
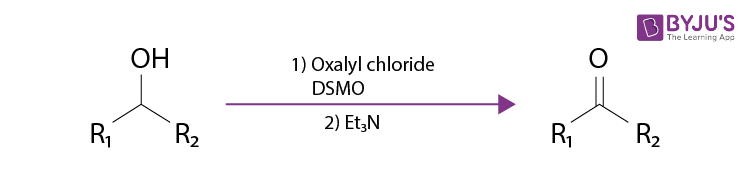
DMSO is the abbreviation used for dimethyl sulfoxide in the example shown above.
Byproducts Produced in Swern Oxidation
The Swern reaction is well known for its wide tolerance range for functional groups as well as its mild characteristics. The following by-products are produced in this type of reaction:
- Dimethyl sulfide (Me2S, also abbreviated as DMS)
- Carbon monoxide (CO)
- Carbon dioxide (CO2)
- Triethylammonium chloride (Et3NHCl, only produced when triethylamine is used as the organic base)
Of the above-given list of byproducts, carbon monoxide and dimethyl sulfide can be considered dangerous. Carbon monoxide is a highly toxic gas to humans since it reacts with the haemoglobin in our blood to produce carboxyhaemoglobin – a compound which is quite ineffective at delivering oxygen to the tissues of the human body.
Dimethyl sulfide is highly volatile in nature. This can be attributed to its boiling point of 37 degrees celsius. DMS has a highly pervasive and strong odour even at low concentrations.
Due to the production of these two compounds as side products, the Swern oxidation reaction must be carried out in a fume hood.
Swern Oxidation Mechanism
Step 1
A resonance-supported dimethyl sulfoxide molecule reacts with oxalyl chloride at low temperatures (temperatures below 600 celsius). An Intermediate is formed and quickly decomposed to give chloro (dimethyl)sulfonium chloride. Carbon monoxide and carbon dioxide are also formed as by-products. This step can be illustrated as follows.
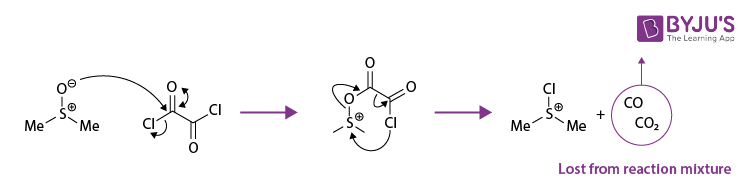
Step 2
Now, an alcohol is added to the chloro(dimethyl)sulfonium chloride. The reaction that ensues gives the required intermediate – the alkoxy sulfonium ion along with hydrochloric acid. This step can be illustrated as

Step 3
Now, a minimum of 2 equivalents of the triethylamine base perform deprotonation of the intermediate alkoxy sulfonium ion. This deprotonation gives the neutral dipolar sulfur ylide. This sulfur ylide now decomposes via a ring transition state which is penta-membered. The products formed from this decomposition include the required carbonyl compound and dimethyl sulfide by-product. This step of the Swern oxidation mechanism is illustrated below.
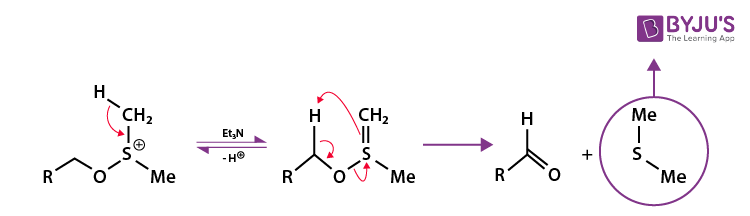
Thus, the required carbonyl compound is achieved from the oxidation of the primary (or secondary) alcohol. Tetrahydrofuran or THF can also be used as a solvent for this reaction.

Comments