Organic reactions are the chemical reactions that are undergone by organic compounds (the chemical compounds containing carbon). A few important types of organic reactions are illustrated below.
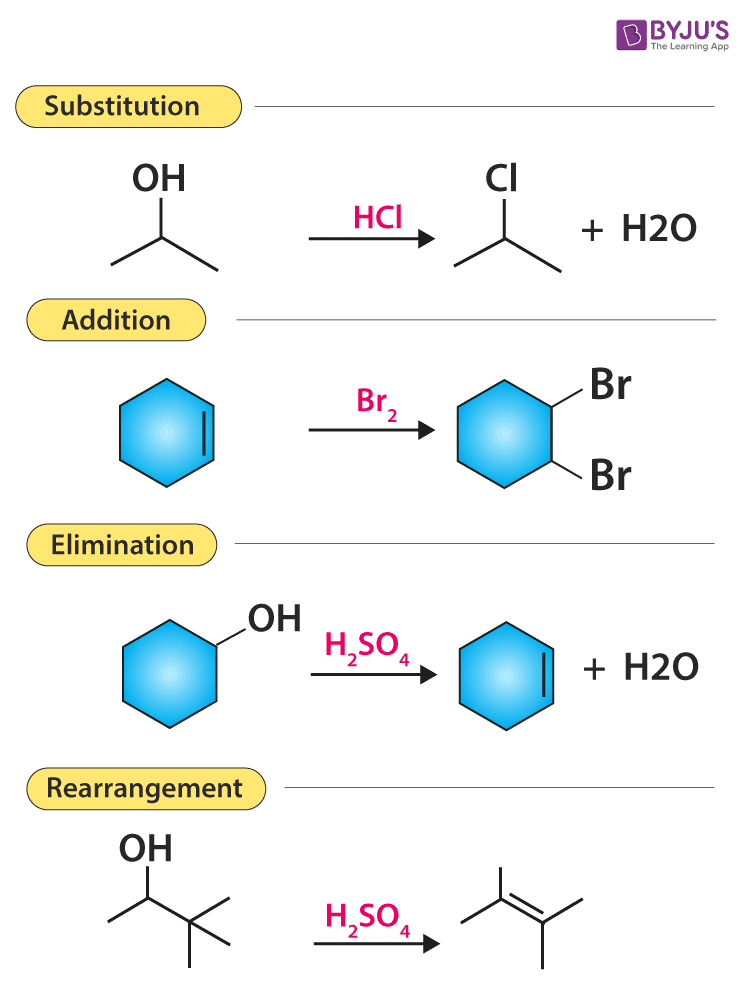
There are mainly five types of organic reactions:
- Substitution reaction
- Elimination reaction
- Addition reaction
- Radical reactions
- Oxidation-Reduction Reactions.
Table of Contents
Substitution Reactions
In a substitution reaction, one atom or a group of atoms is substituted by another atom or a group of atoms to form a new substance.
For example, the conversion of methane to chloromethane is a substitution reaction as hydrogen atoms are replaced by chlorine atoms.
Elimination reaction
There are some reactions which involve the elimination or removal of the adjacent atoms or group of atoms. After this multiple bonds are formed and there is a release of small molecules as products.
One of the examples of elimination reaction is the conversion of ethyl chloride to ethylene.
CH3CH2Cl → CH2=CH2 + HCl
In the above reaction, the eliminated molecule is HCl, which is formed by the combination of H+ from the carbon atom which is on the left side and Cl– from the carbon atom which is on the right side.
Addition reactions
An addition reaction occurs when two or more reactants combine to form a product without the loss of any atoms present in the reactants. Addition reaction is common in compounds that have unsaturated C-C bonds, like double (alkene) and triple (alkyne) bonds. The weaker π bond is converted into two new stronger σ bonds.
HCl + CH2=CH2→ CH3CH2Cl
Also, Read: Redox Reactions
Radical reactions
Many of the organic reactions involve radicals. The addition of a halogen to a saturated hydrocarbon involves a free radical mechanism. There are three stages involved in a radical reaction i.e. initiation, propagation, and termination. Initially when the weak bond is broken initiation of the reaction takes place with the formation of free radicals. After that when the halogen is added to the hydrocarbon a radical is produced and finally, it gives alkyl halide.
To learn more about organic reactions and other important concepts in organic chemistry, register with BYJU’S and download the mobile application on your smartphone.


Comments