Hydrogen formula is discussed in this article. It is non-toxic, highly combustible, nonmetallic, diatomic gas. It is the first element of the periodic table. The molecular formula of Hydrogen is H2.
What Is The Formula Of Hydrogen?
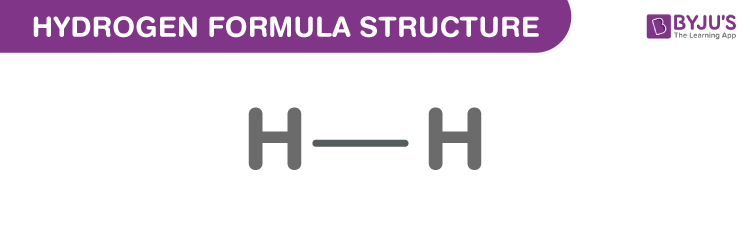
It is a colourless, diatomic gas which is tasteless and odourless compound. Its atomic number is 1. Apart from H1 isotope, hydrogen occurs as Deuterium (stable isotope) and radioactive isotope Tritium (unstable). Dihydrogen consists of two hydrogen atoms joined by a single bond. It acts as an antioxidant, a human metabolite, an electron donor, and a fuel. The structure is given below.
Hydrogen Formula Structure
Properties Of Hydrogen
| Chemical formula | H2 |
| Molecular weight | 2.016 g/mol |
| Density | 0.08988 g/L |
| Boiling point | −252.879 °C |
| Melting point | −259.16 °C |
Use Of Hydrogen
- This compound can easily ignite. On ignition, it burns as a pale blue and invisible flame.
- Hydrogen is widely used in oxyhydrogen cutting, welding, and to make various chemicals.
To learn more about Hydrogen formula from the expert faculties at BYJU’S, register now!
Comments