Ozonolysis is one of the important reactions related to the oxidation of alkenes in organic chemistry. It tells us about the activity of ozone on alkenes. Ozonolysis is sometimes used for detecting the position of double bonds in aliphatic alkenes.
Important Questions For Ozonolysis
Some FAQs related to ozonolysis are underlined below:
Question: What is ozonolysis?
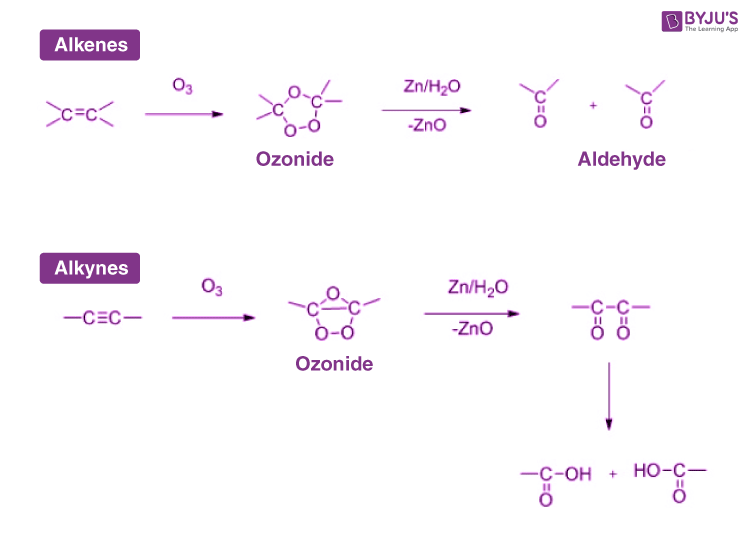
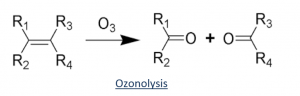
Answer: Ozonolysis is an organic reaction involving the oxidation of alkenes. When a solution containing alkene is exposed to ozone in the presence of an inert solvent, ozonide is formed which on further hydrolysis yields cleavage products like aldehydes and ketones. This reaction is very helpful in the determination of the position of a double bond in aliphatic alkenes.
Question: What is the general mechanism for ozonolysis reaction?
Answer: Ozonolysis reaction generally takes place in two steps:
- Attack of ozone on alkene molecules to form ozonide.
- Formation of cleavage product upon hydrolysis.
A general mechanism is underlined below:
Example Question: A compound on ozonolysis yields ethanal and methanal as the major products. The compound can be:
- Prop-1-ene
- Pentene
- Butylene
- Ethene
Answer: prop-1-ene
Questions: Which reference books can one follow for ozonolysis?
Answer: While covering ozonolysis for JEE, one must go through the NCERT class 11th chemistry textbook part-2. A brisk walk through this topic from NCERT not only gives you a sound idea of the various topics you need to cover according to the JEE syllabus but also develops the fundamentals regarding the chapter. Some authentic books for understanding the mechanism of a reaction include books from authors like Solomons & Fryhle, Morrison & Boyd etc. Once you have developed concepts on this topic, you can practice questions on this topic from the books of authors like MS Chauhan.
For further details related to ozonolysis for JEE chemistry, get in touch with our mentors here at BYJU’S.
Understanding Ozonolysis

Comments