What is Aluminium Phosphate?
Aluminium phosphate is prepared by exposing soluble aluminium salts to alkaline conditions and contains hydrated aluminium orthophosphate with the formula AlPO4.
Table of Contents
- Aluminium Phosphate Structure
- Properties of Aluminium Phosphate
- Physical Properties of Aluminium Phosphate
- Chemical Properties of Aluminium Phosphate
- Uses of Aluminium Phosphate
- Frequently Asked Questions – FAQs
The highly viscous aluminium phosphate solutions tend to form polymeric aggregates, and equilibria are reached only very slowly. It reacts slowly with gastric acid to form soluble aluminium salts and phosphoric acid. It absorbs bile acids less strongly than aluminium hydroxide.
Other names – Aluminium monophosphate
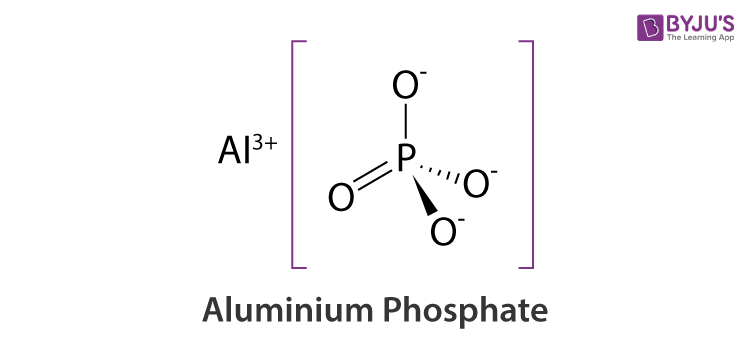
Aluminium Phosphate Structure – AlPO4
Properties of Aluminium Phosphate – AlPO4
| AlPO4 | Aluminium Phosphate |
| Density | 2.57 g/cm³ |
| Molecular Weight/ Molar Mass | 121.9529 g/mol |
| pH | 3.0 to 7.5 |
| Melting Point | 1,800 °C |
| Chemical Formula | AlPO4 |
Physical Properties of Aluminium Phosphate – AlPO4
| Odour | foul odour |
| Appearance | White, crystalline powder |
| Complexity | 36.8 |
| Covalently-Bonded Unit | 2 |
| Hydrogen Bond Acceptor | 4 |
| Solubility | Insoluble in water. |
Chemical Properties of Aluminium Phosphate – AlPO4
- Aluminium phosphate reacts with hydrochloric acid forming phosphoric acid and aluminium trichloride.
AlPO4 + 3HCl → AlCl3 + H3PO4
- Aluminium phosphate reacts with magnesium chloride forming magnesium phosphate and aluminium trichloride.
2AlPO4 + 3MgCl2 → Mg3(PO4)2 + 2AlCl3
Uses of Aluminium Phosphate – AlPO4
- Aluminium phosphate tablets are official in B.P. and each tablet contains 500 mg of dried aluminium phosphate suitable for peppermint flavour.
- Used as an antacid beneficial in patients where significant loss of phosphate occur due to the use of aluminium salts.
- Used in the manufacture of vaccines as an adjunct to enhance immunogenicity
- Aluminium phosphate-carbonate gels were used in water suspension of hydroxy aluminium carbonate with orthophosphoric acid.
Frequently Asked Questions – FAQs
What is the use of aluminum phosphate?
Aluminum Phosphate is an odorless, white crystalline solid which is often used in liquid or gel form. It is used in ceramics, dental cements, cosmetics, paints, paper and pharmaceuticals.
What is the purpose of aluminum in food?
Aluminium-containing food additives have been used in food processing for over a century, as firming agent, raising agent, stabiliser, anticaking agent and colouring matter, etc. and some are permitted to be used in food in many countries. Aluminium is also present in food naturally.
What are the disadvantages of Aluminium foil?
Although aluminum cans are recyclable, they still carry environmental disadvantages, and may pose a health risk to the public. Disadvantages Some disadvantages of using aluminium foil is that the laminated ones cannot be recycled like the normal aluminium foils.
Where does aluminum phosphate come from?
In nature it occurs as the mineral berlinite. Many synthetic forms of aluminium phosphate are known.
Why do we need phosphate?
Phosphate is a charged particle (ion) that contains the mineral phosphorus. The body needs phosphorus to build and repair bones and teeth, help nerves function, and make muscles contract. Most (about 85%) of the phosphorus contained in phosphate is found in bones.

Comments