What is Butyric acid?
C3H7COOH is a short-chain fatty acid with the chemical name Butyric acid. It is also called the Butanoic acid or Propylformic acid or Ethylacetic acid. It is mostly found in its esterified form in plant oils and animal fats. A fatty acid is defined as a carboxylic acid which has a long aliphatic chain, that is either unsaturated or saturated.
It is a colourless oily liquid with an unpleasant odour and has a butter-fat taste. It has a role as a food acidity regulator. It is an ammonium salt and an acetate salt. It is a four-carbon acid. Butanoic acid in its impure form, was first observed by a French chemist the name Michel Eugene Chevreul in the year 1814.
Properties of Butyric acid – C3H7COOH
| C3H7COOH | Butyric acid |
| Molecular weight of C3H7COOH | 88.106 g/mol |
| Density of Butyric acid | 1.135 g/cm3 |
| Melting point of Butyric acid | −5.1 °C |
| Boiling point of Butyric acid | 163.75 °C |
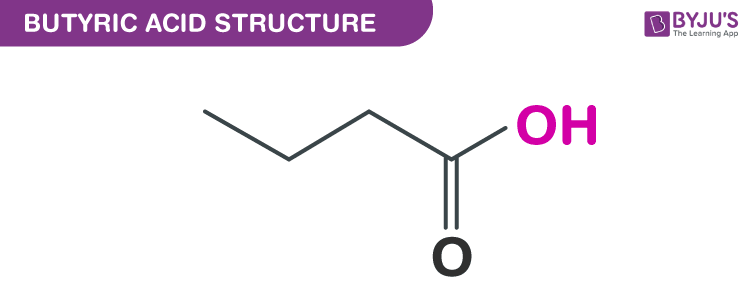
Butyric acid structure – C3H7COOH
C3H7COOH Uses (Butyric acid)
- Butyric acid is used as a flavouring agent in the food industry.
- Used in butter, caramel, and cheese.
- Used in soda water syrups.
- Used in the manufacturing of varnishes.
- Used as an intermediate in industries.
- Used in cosmetics.
- Used in making detergents.
- Used in the production of beer.
- Used in the preparation of esters.
Production of Butyric acid
- Industrially, Ethylacetic acid is prepared by fermenting starch or sugar. It can be more efficient by using Clostridium tyrobutyricum in a catalytic upgrading process. Esters and salts of the acid are called butanoates or butyrates.
- Butyric acid is also obtained in its natural form as the octyl ester from parsnip and from the fruits of Ginko trees.
Health hazards
Inhaling butanoic acid may cause irritation to the mucous membrane, respiratory tract, nausea, and vomiting. Swallowing it causes irritation in the stomach and mouth. When it comes in contact with eyes and skin can cause serious injury and a burning sensation.
Learn more about the Structure, physical and chemical properties of C3H7COOH from the experts at BYJU’S.

Comments