Preparation of Ether – Different Methods
There are various methods for the preparation of Ethers. Ethers are the organic compounds containing an oxygen atom bonded to two same or different alkyl or aryl groups. The general formula for ethers can be R-O-R, R-O-Ar or Ar-O-Ar, where R represents an alkyl group and Ar represents an aryl group.
Ethers are generally classified into two categories on the basis of substituent group attached: symmetrical ethers (when two identical groups are attached to the oxygen atom) and asymmetrical ethers (when two different groups are attached to the oxygen atom). With advancements in technologies, others are synthesized in industries in many ways.
Related Topics
- Diethyl Ether – (C2H5)2O
- Nomenclature of Ether
- Physical and Chemical Properties of Ether
- Uses of Ether
Some ways are explained below.
1. Preparation of Ethers by Dehydration of Alcohols
In the presence of protic acids (sulphuric acid), alcohols undergo dehydration to produce alkenes and ethers under different conditions. For example: in the presence of sulphuric acid, dehydration of ethanol at 443 K yields ethene whereas it yields ethoxyethane at 413 K. This is an ideal method of preparation through primary alcohols.
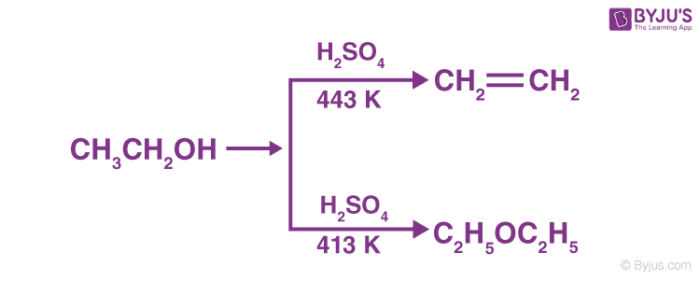
The preparation of ethers by dehydration of alcohol is a nucleophilic substitution reaction. The alcohol involved in the reaction plays two roles: one alcohol molecule acts as a substrate while the other acts as a nucleophile. It can follow either an SN1 or SN2 mechanism. The choice of the mechanism depends on whether the protonated alcohol loses water before or simultaneously upon the attack of a second alcohol molecule. Generally, the secondary and tertiary alcohols follow the SN1 mechanism while the primary alcohols follow the SN2 mechanism.
Recommended Videos
Dehydration of Alcohols and The Mechanisms Behind it

2. Preparations of Ethers by Williamson Synthesis
Williamson’s synthesis is an important method for the preparation of symmetrical and asymmetrical ethers in laboratories. In this method, an alkyl halide is reacted with sodium alkoxide which leads to the formation of ether. The reaction generally follows the SN2 mechanism for primary alcohol.
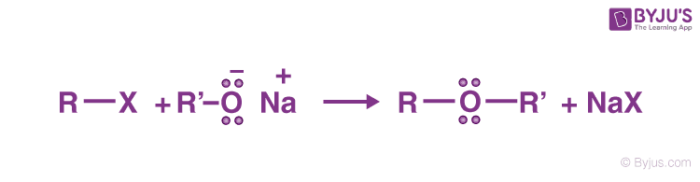
As we know alkoxides are strong bases, and they can react with alkyl halides leading to elimination reactions. Williamson synthesis exhibits higher productivity in the case of primary alkyl halides. In the case of secondary alkyl halides, elimination competes with substitution whereas, we observe the formation of elimination products only in the case of tertiary alkyl halides.
Also, Read ⇒ Classification Of Alcohol, Phenol, and Ether
Frequently Asked Questions – FAQs
Where are ethers used?
Where are ethers found?
How do you identify an ether?
Which ether is more polar in nature?
Are ethers reactive?
For detailed discussions on different methods of preparation of ethers, please visit BYJU’S – The Learning App.

Comments