What is Nature and Characteristics of Matter?
Matter is made up of particles. Matter is not continuous and is particulate in nature. The particles of matter are very small.
Table of Contents
The particle nature of matter has the following characteristics:
- Matter is made up of particles
- Matter has inter particle space
- The particles constituting a matter are very small.
- Particles in matter are always in a state of motion.
- The motion of the particles increases with an increase in temperature.
Nature of Matter
Anything that occupies space and has mass is known as matter. Everything around us is a form of matter. The huge buildings, bridges, electrons revolving around a nucleus, the DNA in our cells, the surrounding air, the land beneath our feet, etc. all matter. A matter is said to be composed of particles which are basically atoms and molecules. Depending upon its physical state, the nature of matter falls into three categories:
-
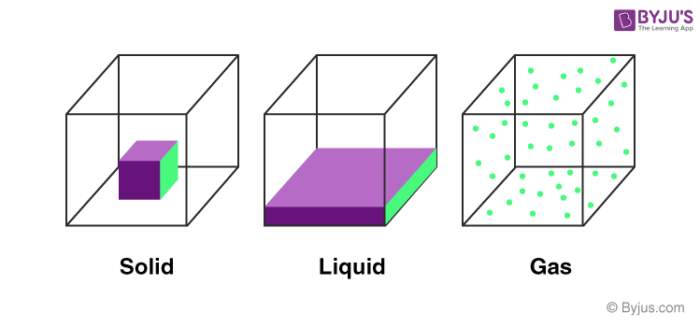
Solids:
The substances in which the particles are held close to each other with strong intermolecular forces are known as solids. The particles are strongly held at their positions and have only vibratory motion. Solids have a definite shape and definite volume. E.g.: Wood, iron, etc.
-
Liquids:
Those substances in which the intermolecular forces are weak enough to allow the movement of particles are known as liquids. The particles are held closely and have a higher degree of freedom than solids. Liquids have a definite volume but no definite shape; they generally take the shape of the container in which they are placed. E.g.: water, milk, etc.
-
Gases:
These types of matter have very weak forces between their molecules and hence the molecules are free to move. The distance between molecules is large as compared to solids and liquids. Gases have neither a fixed shape nor a definite volume. They tend to completely occupy the container in which they are placed. E.g. air, oxygen, hydrogen, methane, etc.
The above three states of matter can be transformed from one form to the other by changing the conditions of temperature and pressure. The nature of matter is also determined by its composition. If the matter is composed of more than one type of particle then it is called a mixture while if it consists of a single type of particles then it is known as a pure substance. Mixtures are further classified as homogeneous and heterogeneous mixtures. Pure substances are also sub-divided as elements and compounds.
The nature of matter continues to be a vast subject of research and recent advancements have revealed other states of matter. Boson-Einstein condensate and plasma are the two other states of matter that have been found recently. To learn more about the three states of matter, register with BYJU’S and download our app.
Frequently Asked Questions – FAQs
What are matter particles?
Matter is composed of elementary particles at the most fundamental level, such as quarks and leptons (the class of elementary particles which includes electrons). Quarks fuse into protons and neutrons and form atoms of the elements of the periodic table, such as hydrogen, oxygen, and iron, along with electrons.
How do particles of matter have spaces?
It is correct to suggest that matter particles have space between them. The physical features of matter suggest that matter has space between its constituent particles. So, it goes into the tiny spaces between water molecules that are present.
How many properties of matter are there?
Four normal forms of matter are present: solids, liquids, gases and plasma. The fifth condition is the Bose-Einstein condensates that are man-made. In a solid, ions are closely bundled together so that they don’t move much.
What are the classification of matter?
It is important to classify matter into two categories: pure substances and mixtures. Elements and molecules are further broken down into basic substances. Mixtures are entities that are mechanically mixed and which can be divided into their original components. One form of atom or molecule is made of a chemical substance.
What is the definition of mixtures?
In chemistry, a combination is a medium made up of physically mixed two or more distinct compounds. In the type of liquids, suspensions and colloids, a mixture is the physical synthesis of two or more substances in which the names are preserved and combined.

Sir wondering classes