What are the Three States of Matter?
The three primary states of matter are the solid, liquid, and gaseous states.
All the materials we see in our daily lives (from ice-cream to chairs to water) are made up of matter. Matter can be classified into different states such as solid, liquid and gas on the basis of intermolecular forces and the arrangement of particles. These three forms of matter can be converted from one state of matter to another state by changing certain environmental factors (increasing or decreasing pressure and temperature, for instance). For example, Ice can be converted from a solid into liquid water by increasing the temperature.
Table of Contents
- Matter Definition
- Recommended Videos
- States of Matter
- Three States of Matter with Examples
- Frequently Asked Questions – FAQs
The state of the matter is also characterized by the transition of phases. The transition process signals a shift in structure and can be detected by a sudden change in properties. A separate state of affairs may be defined as any set of states that are separated from any other set of states by a phase transition.
Matter Definition
Matter can be defined as the material substance that constitutes the observable universe. Matter, along with energy, is known to form the basis of all objective phenomena. In the fields of classical physics and general chemistry, the term matter is used to denote any material that has mass and takes up space by having volume.
Everything that takes up space and has mass is matter, which is the “stuff” that makes up the cosmos. Atoms, which are made up of protons, neutrons, and electrons, are the building blocks of all matter. Chemical energy is a type of potential energy that holds atoms and molecules together. Matter is defined as everything that has mass and volume (takes up space). It is pretty straightforward to illustrate that most everyday items that we deal with on a daily basis have mass and take up space.
Ultimately, all ordinary objects that can be touched are composed of atoms that are made up of interacting subatomic particles, and the term ‘matter’ normally involves atoms and everything made up of them in ordinary as well as a scientific use. Also, any particles (or a mixture of particles) that behave as though they have both residual mass and volume. Massless particles, such as photons, or other energy phenomena or waves, such as light or sound, cannot be referred to as matter.
Recommended Videos

Change in state of matter alters the structure of matter and the arrangement of particles. All of this can be observed by noticing the change in properties.
States of Matter
The condition of the matter is one of the distinct forms that the various phases of the matter take. Four states of matter can be found in daily life: solid, liquid, gas, and plasma. Many other states, such as Bose – Einstein condensate and neutron degenerate matter, are considered to occur only in extreme conditions such as ultra-cold or ultra-dense matter. Other states, such as quark – gluon plasmas, are thought to be possible but remain theoretical for the time being.
The states within the device are in a gaseous, liquid or solid state. Solids are distinguished by a tight atomic bond and a high viscosity, resulting in a rigid form. Most solids are crystalline, in as much as they have a three-dimensional periodic atomic structure; certain solids (such as glass) lack this periodic arrangement and are non-crystalline or amorphous.
The particles (ions, atoms or molecules) are tightly packed together in the solid. The forces between the particles are intense in such a way that the particles can not move freely but can only vibrate. As a result, the solid has a stable, definite shape and a certain volume. Solids can only change their shape by force, as if they were broken or cut.
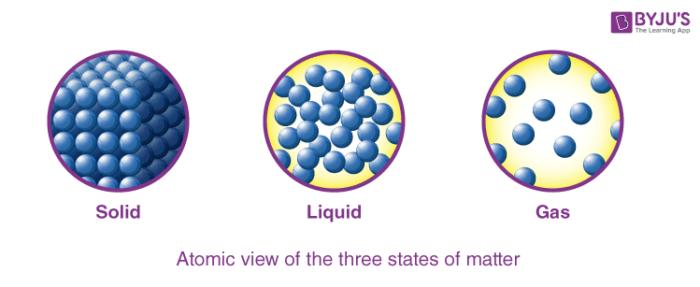
Three States of Matter – Solid, Liquid and Gas
A liquid is an almost incompressible fluid that conforms to the shape of its container but retains a (nearly) constant volume independent of the pressure. Volume is defined if temperature and pressure are constant. As the solid is heated past its melting point, it becomes liquid as the pressure becomes greater than the triple point of the material.
Within a gas, the molecules have enough kinetic energy such that the impact of the intermolecular forces is small (or zero for the ideal gas) and the normal distance between the adjacent molecules is much greater than the molecular size. The gas has no definite shape or volume, but it occupies the entire container in which it is confined.
Three States of Matter with Examples
There are three states of matter and below are the description of various states of matter:
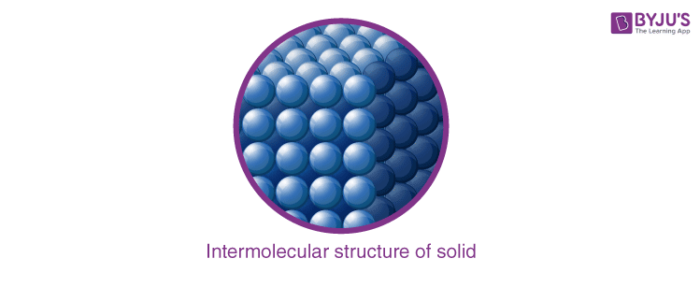
1. Solids
- The solid state is one of the fundamental states of matter.
- Solids differ from liquids and gases by the characteristic of rigidity.
- The molecules of solids are tightly packed because of strong intermolecular forces; they only oscillate about their mean positions.
- Whereas, liquids and gases possess the property of fluidity and can easily flow.
- Solids can be defined as the state of matter which has definite shape and volume and has a rigid structure.
- Solids possess the least compressibility and thermal expansion.
Example: Iron (Fe)
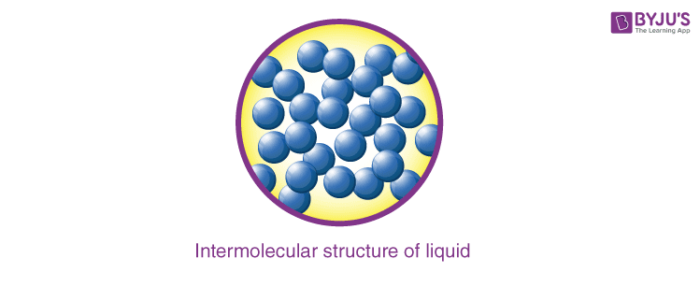
2. Liquids
- The molecules in a liquid are closely packed due to weak intermolecular forces.
- These forces are weaker than solids but stronger than that of gases.
- There is much space in between the molecules of liquids which makes their flowing ability easy.
- Liquids can easily acquire the shape of a vessel, and they have a fixed volume.
- Conversion of solids into liquids takes place when we increase the temperature of solids to a point where solids begin to melt.
- Generally, the density of liquid lies between the density of solids and gases. Compressibility and thermal expansion of liquids are slightly higher than that of solids.
Example: Water (H2O)
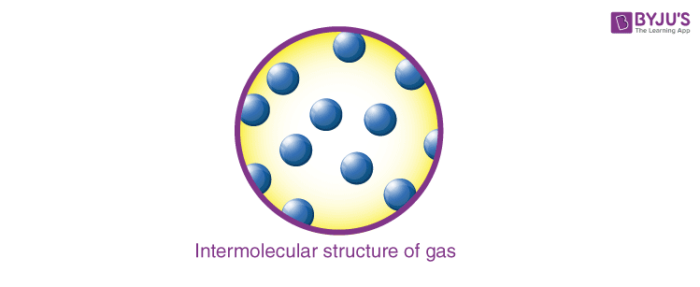
3. Gases
- In this state of matter, distances between the molecules are large (intermolecular distance is in the range of 10-7 – 10-5 cm).
- The intermolecular forces experienced between them are negligible.
- Thus, translatory, rotatory and vibratory motions are observed prominently in gases.
- Gases do not have any fixed shape or volume.
- They also possess high compressibility and thermal expansion.
Example: Oxygen (O2)
Frequently Asked Questions – FAQs
Can Matter exist in two states at once?
Pressure can turn matter from one state to another. Certain matters often vary, but often only occurs in two states and requires human and technical help to travel through all three stages. Water is the only thing on earth that can naturally be present in all three-solid, liquid and oil.
How do you explain matter?
Matter is all around you. All atoms and compounds are made up of very small pieces of matter. Such atoms continue to build the everyday things you see and touch. Matter is defined as anything that has mass (it has volume) and takes up space.
Why are the 3 states of matter important?
Three types of matter are solids, liquids, and gases. Comprehending the particle nature of matter is significant. Particles that makeup matter are not ‘ small solid bits ‘ or ‘ small liquid drops, ‘ but atoms and molecules. Such atoms and molecules ‘ physical characteristics determine their state.
How is the matter made up?
Matter is also composed of small particles. Atoms are the particles that makeup matter. Because they are so small you can’t see atoms. Most atoms come together to make up the substance you can see.
Is lights a matter?
Light is, not matter, a form of energy. Matter consists of atoms. Actually, light is electromagnetic radiation. Moving electrical charge or electrons (electrical current) causes a magnetic field and a changing magnetic field creates an electrical or electrical field.
What are the states of matter liquid?
A liquid is an almost incompressible fluid that conforms to the shape of its container but retains a (nearly) constant volume independent of the pressure. As such, it is one of the four fundamental states of matter (the other being solid, gas, and plasma) and it is the only state with a definite volume but without a fixed form.
How do we classify matter?
Matter can be categorized by physical and chemical properties. Matter is something that takes up space and has a mass. The three states of matter are solid, liquid and gaseous. Physical transition includes the conversion of a material from one state of matter to another without changing it’s chemical composition.
How many types of matter are classified?
The three states of matter are the three distinct physical forms which matter can take in most environments: solid, liquid , and gas. Other states, such as plasma, Bose-Einstein condensate, and neutron stars, may be present in extreme environments.
In this article, we have studied the three states of matter. To know more about the states of matter, download BYJU’S – The Learning App.

really helpful for students