What is DDT?
DDT stands for dichlorodiphenyltrichloroethane. It is a colourless, tasteless and almost odourless organochloride. DDT is popularly known for its insecticidal properties. It has been easily formulated in almost every form which includes solutions in petroleum distillates, emulsifiable concentrates, and smoke candles.
Table of Contents
Earlier Uses of DDT
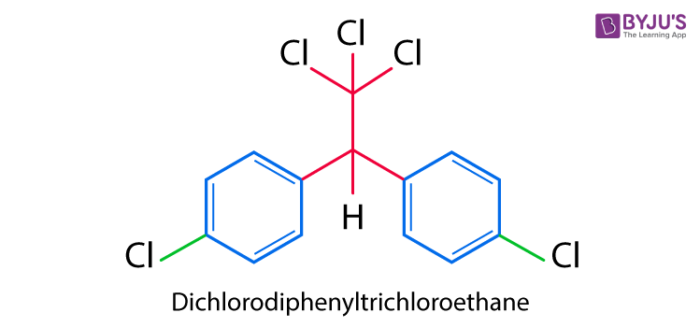
DDT is the first chlorinated organic insecticide and was prepared in the year 1873. Paul Muller of Geigy Pharmaceuticals in Switzerland was the first person to discover the effectiveness of DDT as an insecticide. It was largely used after the Second World War to control malaria and typhus among the civilians and the troops. After the war, the chemical was made available for agricultural uses and therefore its use immensely increased over time. The structure of DDT is as follows:
Problems
The problems related to extensive use of dichlorodiphenyltrichloroethane began to appear in the late 1940s. Many insects developed resistance towards it and later it was found that the chemical had toxic effects towards fishes. The chemical stability and fat solubility property of DDT compounded this problem.
In insects, DDT opens the sodium ion channel which is present in the neutrons. This causes a spontaneous fire which leads to spasms and eventually death. There are certain insects with a certain type of mutation in their sodium channel gene that are resistant to DDT. Animals do not metabolize it rapidly, instead they deposit and store the chemical in their fatty tissues. If this ingestion continues, dichlorodiphenyltrichloroethane is build up within the animals over time.
Scientists started expressing their concern over the hazards associated with DDT. The government tightened the laws that governed its usage. Many environmentalists argued that pesticides including dichlorodiphenyltrichloroethane had a poisoning effect on both wildlife and the environment.
It has endangered human health. Bird killing or decline in the bird population resulted due to an adverse impact of this chemical. Therefore DDT was banned in the USA. It is still used in some parts of the world, but its use should be prohibited to sustain a healthy life and environment.
This article deals with the properties, uses, and effects of dichlorodiphenyltrichloroethane. To learn more about this concept, download BYJU’S – The Learning App.

Comments