What is Swarts Reaction?
Swarts’ reaction is generally used to get alkyl fluorides from alkyl chlorides or alkyl bromides. This is done by heating of the alkyl chloride/bromide in the presence of the fluoride of some heavy metals (silver fluoride or mercurous fluoride for example). The reaction will proceed if sodium fluoride or potassium fluoride is used, but the resulting yield will be significantly lower. This process was first reported by Frederic Jean Edmond Swarts in 1892.
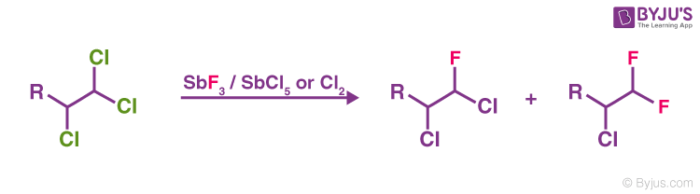
In Swarts fluorination, chlorine is most commonly replaced by fluorine in organic compounds with the help of antimony trifluoride in the presence of antimony salts where its oxidation state is +5. A few examples of this type of reaction can be found below:
The Swarts reaction mechanism is quite simple – the metal fluorine bond is broken and a new bond is formed between carbon and fluorine. The displaced chlorine or bromine atoms now bond with the metal. The mixture of antimony trifluoride and chlorine is referred to as Swarts reagent. Swarts rule states that post fluorination, the fluoride formed will always have a lower boiling point than the corresponding chloride.
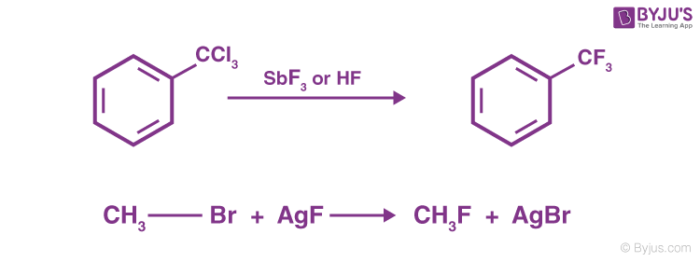
Chlorinated hydrocarbons can also be reacted with metallic fluorides to give hydrocarbons with chlorine and fluorine attached. For example:

Thus, Swarts fluorination can be used for the complete replacement of chlorine or bromine with fluorine in alkyl chlorides/bromides. A variant of the reaction is quite important in the production of Freons. In this variation, the fluorination is executed using anhydrous hydrogen fluoride in the presence of antimony salts (of catalytic quantities) where antimony displays oxidation levels of +3 and +5.
Frequently Asked Questions – FAQs
What is Swarts reaction explain with example?
In general, Swarts’ response is used to extract alkyl fluorides from alkyl chlorides or alkyl bromides. This is achieved in the presence of the fluoride in certain heavy metals by boiling the alkyl chloride / bromide.
What are Finkelstein and Swarts reaction?
The reactions of Swarts and Finkelstein are halogen exchange reactions related to alkyl halides. In this reaction, sodium iodide (the nucleophile) is treated with ethyl chloride (a main alkyl halide) to produce ethyl iodide.
Which reagent is used in Swarts reaction?
In the presence of antimony pentachloride or chlorine, the partial fluorination of nonpolar organic polyhalides (mostly aliphatic) with antimony trifluoride is usually referred to as the Swarts reaction and the combination of antimony trifluoride and chlorine (SbF3 + Cl2) is referred to as the Swarts reagent.
What is retention in configuration?
A system in which the atom’s relative configuration or absolute configuration is preserved. If the atom in question is a stereo centre, structure retention typically (but not always) adjusts the absolute configuration of R to R, and S to S.
What is the product of Finkelstein reaction?
An SN2 reaction in which another halogen atom (the nucleophile) replaces one halogen atom (the leaving group). In this example of the Finkelstein reaction, sodium iodide (the nucleophile) is treated with 1-chloro-2-phenylethane (a main alkyl halide) to create 1-iodo-2-phenylethane.

Comments