Rajasthan 10th Standard General Science Exam Question Paper 2017 with Solutions – Free Download
Rajasthan Board Class 10 General Science 2017 Question Paper Solutions here are very suited for the students to prepare for exams. These solutions are the necessary foundation for the important questions that can most likely be found in the Class 10 General Science exam. Knowing the entire concepts and topics from the textbooks of the respective subject will help the students to solve these RBSE Class 10 Science 2017 Previous Year Paper. Referring to the solution after will also help them to gauge their exam preparation levels.
To get these PDFs of solved or unsolved RBSE 10th question paper, students have to use the respective links listed below. These clickable PDF links listed here are interactive. Students can download printable formats of the question papers from these links. Meanwhile, the questions and answers are also published on the web page.
Students are urged to familiarise themselves with the complex topics from the Class 10 General Science textbook. These solutions will help the students to get an overview of the question paper design of the RBSE Class 10 General Science exams. They are advised to solve the previous papers of RBSE Class 10 General Science while preparing for the exams. Practising these papers and referring to solutions will help students to self gauge their performance and preparation level for the final exams. If the students are regular in solving these paper solutions, they will also get an idea about the marks’ distribution in each section.
Download Rajasthan Board 10th 2017 General Science Question Paper
Download Rajasthan Board SSLC 2017 General Science Solutions
Rajasthan Board Class 10 General Science 2017 Question Paper with Solutions
Part- A
- Write two ways to prevent rusting of iron.
Answer: Iron can be prevented from rusting, by coating it with paint or varnishing the surface. Galvanisation is the other method used to prevent rusting.
2. 2Mg+ O2 → ?
Complete the above reaction.
Answer: 2Mg+ O2 → 2MgO
3. Write two disadvantages of drinking alcohol in excess.
Answer: Drinking alcohol in excess could result in loss of self-control, cause damage to the liver like liver cirrhosis or result in nerve damage. Excessive drinking can also lead to obesity.
4. Write two isotopes of chlorine.
Answer: Cl-35 and Cl-37 are the stable isotopes of chlorine.
5. Write the name of a Parasite, which causes Malaria disease.
Answer: Malaria is a mosquito-borne infectious disease caused by various species of the parasitic protozoan microorganisms called Plasmodium.
6. Write the value of the ‘least distance of distinct vision’ for a young adult with normal vision.
Answer: The value for ‘least distance of distinct vision’ for a young adult with normal vision is 25 cm.
7. In the given ray diagram write the value of angle of incidence and the name of refracted ray,

Answer: In optics, angle of incidence can be defined as the angle between a ray incident on a surface and the line perpendicular to the surface at the point of incidence (called as normal). Here, the angle of incidence will be 90° – 30°= 60°. P is the refracted ray from this figure.
8. Write the equivalent resistance of the given circuit

Answer: The circuits here are connected in series so the equivalent resistance will be, R = R1 + R2 + R3 = 1 + 2+ 3 = 6. Hence, the equivalent resistance of the given circuit is 6 Ω.
Part-B
9. Differentiate between calcination and roasting.
Answer: Calcination is a process in which ore is heated in the absence of air or air might be supplied in limited quantity. Alternatively, Roasting involves heating of ore lower than its melting point in the presence of air or oxygen.
Find here the difference between calcination and roasting.
10. (a) Write names of any two conventional sources of energy.
(b) Write any two limitations in harnessing wind energy.
Answer: (a) The conventional sources of energy include coal, oil and Natural Gas, Electricity and so on.
(b) The limitation of harnessing wind energy is given here. A windmill requires a wind of speed more than 15 km/h to generate electricity from wind energy also large numbers of windmills are required to get a feasible output which covers a large area. The cost of setting up the windmills is also high.
11. If long plants of pea with round seeds are crossed with short plants having wrinkled seeds, which types of plant will be obtained in the first and the second generations? Write the phenotypic ratio of plants obtained is a second generation(F2).
Answer: The progeny created by crossing a long pea plant having round seeds (TTRR) with a short plant having wrinkled seeds (ttrr) are all long plants with round seeds (TtRr). That is because, here, tallness and round shape were the dominant traits, while wrinkled and short seeds are recessive traits. It is seen that when F1 progeny is cross-bred using self-pollination, then 4 different types of progeny are produced in the F2 generation. Of these, it is seen that two types will have the same trait as the parents and the other two will have a combination. So,
Tall plants and round seeds -9
Tall plants and wrinkled seeds-3
Short plants and round seeds-3
Short plants and wrinkled seeds-1
Hence, the phenotypic ratio in the F2 generation will be- 9:3:3:1.
12. What is the basis of sex-determination in human beings? Explain.
Answer: Human beings have 23 pairs of chromosomes, all of which are paired except for the sex chromosomes that are odd and not perfect. Women have a perfect set of sex chromosomes, i.e. the XX, while men have a normal-sized ‘X’ chromosome and a short-sized ‘Y’ chromosome forming XY chromosome. Hence, all children will inherit ‘X’ chromosome from their mother regardless of them being a girl or a boy. Moreover, from their father, they inherit either ‘X’ or ‘Y’. This means the sex of a child is determined by the chromosome, they inherit from the father.
13. (a) Write the name of a spherical mirror used as a rearview mirror.
(b) An electric bulb is connected to a 220 V generator. Write a value of the power of the bulb if 0.5 A current is flowing through the bulb.
Answer: (a) The spherical mirror used as a rearview mirror is the convex mirror.
(b) Here, apply the formula P= VI = 220 x 0.5 = 120 watt. So,the value of the power of the bulb is 120 watt.
14. Why are in the household electric circuits, the electric appliances (instruments) connected in parallel combination?
Answer: Electric appliances connected in parallel combination can be switched on and off, independently. Also, the same voltage from the main lines is available for all the electric appliances.
Part-C
15. (a) What is done to prevent the rancidity in foods containing fats?
(b) CuO + H₂ → Cu + H2O
In the above reactions, which substance is being oxidised and which is reduced?
Answer: (a) Often the rancidity in food containing fats is due to oxidation. So, antioxidants are added to prevent the rancidity. Storing the food in airtight containers or storing in the refrigerator is another way to prevent rancidity.
(b) In oxidation, as the species lose electrons, the oxidation number increases. Alternatively, in reduction, the species gain one or more electrons, and the oxidation number is reduced. Hence, here the hydrogen is oxidised, and copper is reduced. So, hydrogen is known as the reducing agent, while copper(II) oxide is known as the oxidising agent.
16. (a) What are isotopes?
(b) State modern periodic law.
(c) Name two noble gases.
Answer: (a) Isotopes of an element have the same physical properties, but different chemical properties. They are elements with the same atomic number and different mass number. Meanwhile, their number of protons is the same but neutrons are different.
(b) Physical and chemical properties of elements are a periodic function of their atomic numbers (or protons). Modern periodic law states that the properties of elements are dependent on their atomic numbers and elements show similar properties at a regular interval of increasing atomic number.
(c) Helium and Neon are examples of noble gases.
17. (a) Write two properties of metals.
(b) What is an alloy? Give two examples.
(c) What is the nature of oxides of metals and non-metals?
Answer: (a) Metals are minerals or substances that form naturally below the surface of the Earth. Most metals are lustrous or shiny. They are also strong and durable.
(b) An alloy is a homogeneous material that can be created by melting two or more elements, of which one is a metal. Participatory metal is known as parent metal, which serves as a base metal for the alloying element and is considered alloying agents for the majority of the substances. The alloying agents can either be non-metal or metal in which their contribution or proportion is minimal. Alloys tend to have better/more desirable properties when compared to the properties of the constituent substance they are made of. Almost all the alloys are lustrous (which can be explained by the presence of the metallic component in their composition). Steel, brass, bronze, etc. are a few examples of alloys.
(c) Oxides of metals are basic as they create salt and water when dissolved in water. They also contain OH- ions. Meanwhile, the oxides of non-metals are acidic.
18. (a) Explain the role of enzymes in the process of digestion in human beings briefly.
(b) In view of road safety, what is the permissible level of alcohol in 100 ml of human blood, legally?
Answer: (a) Chemical processes are achieved by the following enzymes and hormones, Salivary Aamylases and Lysozymes. The oral cavity produces these enzymes. The primary function of these enzymes is to split the carbohydrates by the hydrolytic actions. The functions of these digestive enzymes are as follows:
- Salivary amylases – 30% of starch molecules are converted into maltose by salivary amylases at the pH of 6.8.
- Lysozymes – It acts against bacterial infections. They are also called antibacterial agents.
(b) In India, the Motor Vehicle Act states that 0.03% or 30 mg per 100ml of blood in humans is permissible.
19. Write the name and function of hormone secreted by Pancreas gland. Write the name of the disease caused due to its deficiency.
Answer: Pancreas gland secretes hormones such as Glucagon, Insulin, Gastrin, Somatostatin and Vasoactive Intestinal Peptide (VIP). Insulin controls the metabolism of carbohydrates, and its deficiency could result in diabetes mellitus. Glucagon helps in the release of sugar, thus increasing the blood sugar levels. VIP controls the secretion of water and the intestinal absorption, while Gastrin helps with digestion of food. Finally, somatostatin maintains the glucose and the salt balance in the food.
20. Draw a labelled diagram of a human brain and write any of its two functions.
Answer: The human brain controls nearly every aspect of the human body ranging from physiological functions to cognitive abilities. It functions by receiving and sending signals via neurons to different parts of the body. The human brain, just like most other mammals, has the same basic structure, but it is better developed than any other mammalian brain. The brain, along with the spinal cord, constitutes the central nervous system. It is responsible for thoughts, interpretation and origin of control for body movements. The human brain consists of the forebrain, midbrain and the hindbrain. Forebrain controls the reproductive functions, body temperature, emotions, hunger and sleep. Alternatively, the three regions of the hindbrain coordinate all processes necessary for survival. These induce breathing, heartbeat, sleep, wakefulness and motor learning.

21. What will happen if the placenta is damaged during the development of an embryo? Explain.
Answer: During the development of an embryo, if the placenta gets damaged, then the adequate supply of oxygen and minerals from the mother to the baby will be stopped. If this important support malfunctions then the baby will not develop properly, resulting in low birth weight, premature birth and other defects. Placenta damage can also be dangerous for the mother causing complications. Hence, it is required to diagnose this problem as early as possible.
22. (a) If a person cannot see nearby objects, then write the name of the eye defect. Write the name of the lens used to correct this eye defect.
(b) In brief, define the Tyndall effect.
Answer: (a) Hypermetropia is the eye defect where a person can see faraway objects clearly and distinctly, but they are not able to see nearby objects comfortably. Convex lenses are used to correct this eye defect, as it can converge the incoming rays.
(b) The Tyndall effect is the phenomenon in which the particles in a colloid scatter the beams of light that are directed at them. This effect is exhibited by all colloidal solutions and some very fine suspensions.
23. Write Ohm’s law. Write the name of devices X and Y in the given circuit, related to Ohms.
 .
.
Answer: Ohm’s law states that the voltage across a conductor is directly proportional to the current flowing through it, provided all physical conditions and temperature remain constant. It shows the relationship between electric current and potential difference. The current that flows through most conductors is directly proportional to the voltage applied to it. Mathematically, this current-voltage relationship can be written as, V=IR. In the equation, the constant of proportionality, R is Resistance and has units of ohms, with symbol Ω. In this image, the device X is the ammeter, while Y is the voltmeter.
24. Define electric current. A current of IA is drawn by a filament of an electric bulb for 30 seconds. Find the amount of electric charge that flows through the circuit.
Answer: The rate of flow of negative charges of a conductor is electric current. It is also known as the continuous flow of electrons in an electric circuit. Conducting material consists of numerous free electrons that randomly move from one atom to the other. Here, I=qt , so q= it.
Therefore,q= 1×30=30C
Thus, the amount of electric charge that flows through the circuit is 30 C.
Part- D
25. (i) What is the effect of hydrogen ion[H+] concentration on the nature of the solution?
(ii) When acid is diluted, acid is added in water, not water in acid. Give reason.
(iii) Name the acid present in Orange and Tamarind.
(iv) What is the chlor-alkali process?
Answer: (i) The concentration of the hydrogen ion[H+] determines that the solution is acidic. So, with the increase in the concentration of hydrogen ion[H+], the acidic nature of the solution also increases or with the acidic nature, the hydrogen ions increase.
(ii) The reaction of water with the acid, is an exothermic reaction. So, if the water is added to the acid, chances are there of explosion or sudden splashes, thus injuring someone. So, to dissipate the heat and to avoid further danger, acid is added to the water.
(iii) The acid present in the orange is the citric acid and the tartaric acid in the tamarind.
(iv) Learn what is the chlor-alkali process.
26. (a) What is a detergent?
(b) Explain the causes of ozone depletion.
(c) Draw a labelled diagram of ‘Khadin water harvesting system’ and write any two measures of water conservation.
Answer: (a) Detergents are the potassium or sodium salts of a long alkyl chain ending with a sulfonate group and are soluble in hard water. This solubility is attributed to the fact that the sulfonate group does not attach itself to the ions present in hard water. Commonly, anionic detergents such as alkyl benzene sulfonates are used for domestic purposes.
(b) The ozone layer depletion is a significant concern and is associated with various factors. The main causes responsible for the depletion of the ozone layer are listed below:
Chlorofluorocarbons
Chlorofluorocarbons or CFCs are the main cause of ozone layer depletion. These are released by solvents, spray aerosols, refrigerators, air-conditioners, etc. The molecules of chlorofluorocarbons in the stratosphere are broken down by the ultraviolet radiations and release chlorine atoms. These atoms react with ozone and destroy it.
Unregulated Rocket Launches
Researchers say that the unregulated launching of rockets result in much more depletion of the ozone layer than the CFCs do. If not controlled, this might result in a huge loss of the ozone layer by the year 2050.
Nitrogenous Compounds
The nitrogenous compounds such as NO2, NO, N2O are highly responsible for the depletion of the ozone layer.
Natural Causes
The ozone layer is depleted by specific natural processes such as Sun-spots and stratospheric winds. Nevertheless, it does not cause more than 1-2% of the ozone layer depletion. The volcanic eruptions are also responsible for the depletion of the ozone layer.
(c) Here are two measures used to conserve water:
- Tightly close the piper after use
- Repair the pipe leakages
- Use high-efficiency washing machines
27. (a) Draw a labelled diagram of an electric generator. Explain its construction and working in brief.
(b) Write down any two characteristics of magnetic field lines of a bar magnet.
Answer: (a) Find here the diagram of the electric generator along with its working.
(b) Given here are some characteristics of the magnetic field lines of a bar magnet:
- The magnetic field lines of a magnet form continuous closed loops and do not intersect
- The tangent to the field line at any point represents the direction of the net magnetic field B at that point.
28. (a) Explain with an example:
(i) Saturated hydrocarbon
(ii) Hydrogenation of unsaturated hydrocarbons
(b) Write two characteristics of the carbon atom.
(c) What is the substitution reaction?
Answer: (a) (i) A Saturated hydrocarbon is a hydrocarbon in which all the carbon-carbon bonds are single bonds. A hydrocarbon is an organic compound whose only constituents are carbon and hydrogen. As the name suggests, saturated hydrocarbons are hydrocarbons in which all the carbon atoms are bonded to four other atoms and are ‘saturated’, implying that no carbon-carbon multiple bonds exist in these organic compounds. Generally, the term ‘saturated hydrocarbon’ is used to refer to alkanes – acyclic hydrocarbons containing only sp3 hybridised carbon atoms. The general formula of an alkane is CnH2n+2. Butane and Octane are an example of saturated hydrocarbons.
(ii) A reaction in which hydrogen molecules are added to an alkene is called a hydrogenation reaction also it is called a reduction reaction.
(b) Some characteristics of carbon is as given below:
- The atomic number of carbon is 6, and the atomic mass is 12.01gmol-1.
- Represented by the symbol C and present in the 14th group of elements in the Periodic Table.
- It is available as coal and graphite in the elemental state.
(c) The substitution reaction is defined as a reaction in which another group substitutes the functional group of one chemical compound, or it is a reaction which involves the replacement of one atom or a molecule of a compound with another atom or molecule.
Or
- Differentiate between soap and detergents.
- Why is pure acetic acid called glacial acetic acid?
- What is a covalent bond? Write two properties of covalent compounds.
Answer: (a) Learn here about the differences between the soap and detergent.
(b) Glacial acetic acid is a pure or concentrated form of acetic acid and is also called anhydrous acetic acid, as it contains less than 1% water. Glacial acetic acid is also colourless and is very corrosive. Glacial acetic acid also forms ice-like crystals, at just below room temperature.
(c) Elements having very high ionisation energies are incapable of transferring electrons and elements having very low electron affinity cannot take up electrons. The atoms of such elements tend to share their electrons with the atoms of other elements or with other atoms of the same element in a way that both the atoms obtain octet configuration in their respective valence shell and thus achieve stability. Such association through sharing of electron pairs among different or same kinds is known as Covalent Bond. A covalent bond is formed by equal sharing of electrons from both the participating atoms. The pair of electrons participating in this type of bonding is called a shared pair or bonding pair. The covalent bonds are also termed as molecular bonds. Sharing of bonding pairs will ensure that the atoms achieve stability in their outer shell which is similar to the atoms of noble gases. Meanwhile, the characteristics of covalent bonds are as given below:
- Most compounds having covalent bonds exhibit relatively low melting points and boiling points.
- Compounds with covalent bonds usually have lower enthalpies of vaporisation and fusion.
29. (a) Draw a labelled diagram of a human heart.
(b) What is the use of platelets in maintenance in human beings?
(c) Which respiratory pigment is found in red blood corpuscles present in human blood?
Answer: (a)
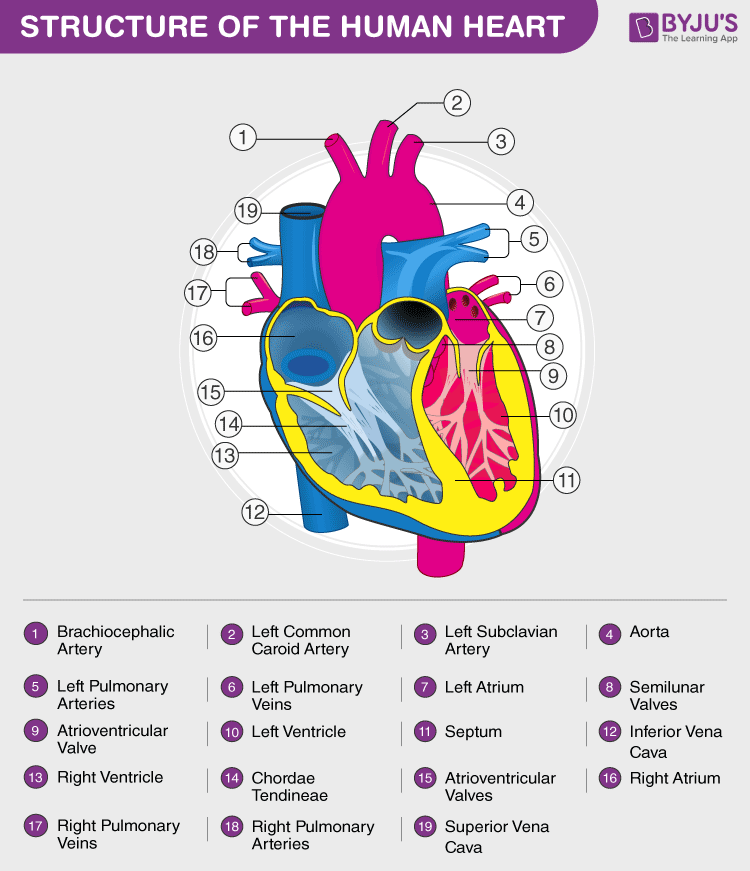
(b) Platelets are also known as thrombocytes and are specialized blood cells produced from bone marrow. The platelets come into play when there is bleeding or haemorrhage. They help in clotting and coagulation of blood. Platelets help in coagulation during a cut or wound.
(c) Haemoglobin, present in human blood, is the respiratory pigment found in the human respiratory system. It is in red colour and is very important for human survival as it carries dissolved oxygen.
Or
- What is lymph?
- Draw a labelled diagram of a human excretory system.
- How plants excrete waste products. Explain.
Answer: (a) Lymph is a clear to a pale-white fluid which circulates throughout the lymphatic system. The main role of the lymphatic system is to act as a filter against microbes, organic wastes and other debris. The lymph comprises lymph plasma, lymph corpuscles and lymphoid organs.
(b)

(c) Elimination of toxic and waste products from the body is called excretion. Learn more about excretion in plants.
30. (a) Define the principal focus of a convex lens. Draw a light ray diagram of an image formed, when the object is placed in between principal focus F and 2F.
(b) Write the lens formula.
(c) Calculate the power of a lens if its focal length is 0.5 m.
Answer: (a) The principal focus of a convex lens is defined as the point, on which the principal axis of the light is parallel to a principal axis that converges after the refraction from the lens.

(b) Find details about Lens Formula.
(c) Given that focal length is 0.5 m, then the Power of convex lens,
P = 1/F= 1/0.5 = 0.5. So, the power of convex lens is 0.5 dioptre
Or
- Define the centre of curvature of a spherical mirror.
- Draw a ray diagram for the image formation by a concave mirror when the size of an object and its image are the same.
- Write the mirror formula.
- Calculate the speed of light in a medium if a refractive index of the medium is 1.5 and the speed of light in a vacuum is 3 x 108 m/s.
Answer: (a) The point in the centre of the mirror that passes through the curve of the mirror with the same tangent and curvature at that point is the centre of curvature of a spherical mirror.
(b) In this, the object is placed at C.

(c) Learn about mirror formula.
(d) Given that n is the refractive index and c is the velocity of light in a vacuum ( 3 × 108 m/s), then v is the velocity of light in a substance.
Now given that n= 1.5 and c = 3 x 108, then
n = c/ v
So, v = c /n = (3 x 108) / 1.5 = v = 2 × 108 ms-1.











Comments