What are Nitro Compounds?
While studying Chemistry, most of us would have come across the term named nitro compounds, and students would be naturally curious to know more about what they are. The organic compounds that have one or more groups that are nitro functional (-NO2) are called nitro compounds.
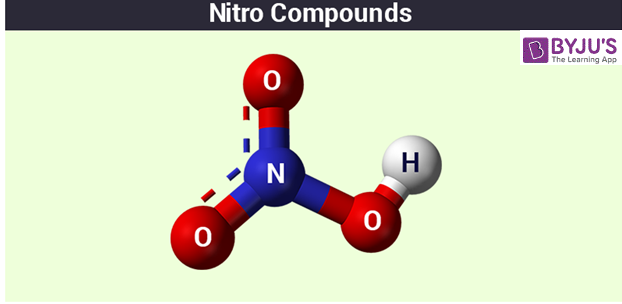
Simply put, any group of chemical compounds that has the nitro group (-O-N=O) becoming a member of the structure of molecules is a nitro compound. Certain examples include organic substances that have the covalent bond of a nitrogen atom of the nitro group connected to an atom of carbon. These compounds have a polar nature, and the ones having no chemically reactive group of their own usually have no colour or either would have a pale yellow colour that would dissolve in the water quite easily. These compounds are mainly used in producing things like explosives, chemical intermediates or solvents.
Formation of the nitro compound
The reaction named nitration by the mixing of an organic compound and nitric acid helps in the formation of the nitro compound. Toluene or benzene, which are the aromatic compounds in nitration, is affected usually when they are treated with a mix of sulphuric and nitric acids at 1000c or less temperature. The notable fact about this is that such temperatures would not be helpful for nitrating aliphatic compounds. However, in the case of propane, it could be used for commercial purposes like converting the mixture of 1-nitropropane, nitroethane, 2-nitropropane, and nitromethane by causing the reaction at 4000C temperature with nitric acid vapours. The fractional distillation would be used to separate the components of this mixture. A wide array of agents causes the reduction of aromatic compounds. The amine is produced by reduction mostly owing to the acidic conditions. Hydroxylamine is produced by reduction when the conditions are neutral. Nitrogen to nitrogen bond compounds is formed in an alkaline solution.
To quote a few examples of natural occurrences of this chemical, we have one in the form of chloramphenicol, and there are some nitro groups formed by amino groups by oxidation. Thus the occurrences of these compounds in nature are really rare.
Thus from the above discussion, we would have realized why nitro compounds are discussed among students owing to their important status in the domain of Chemistry and would continue to do so in the years to come.
To know more about this topic in a detailed and interesting way, download Byju’s-the learning app.

Comments