Table of Contents
What is the Chemistry Behind the Rusting of Iron?Why is Rusting an Undesirable Phenomenon?Factors that Affect the Rusting of IronHow can Rusting be Prevented?
Rusting of iron refers to the formation of rust, a mixture of iron oxides, on the surface of iron objects or structures. This rust is formed from a redox reaction between oxygen and iron in an environment containing water (such as air containing high levels of moisture). The rusting of iron is characterized by the formation of a layer of a red, flaky substance that easily crumbles into a powder.

Rusting of Iron
This phenomenon is a great example of the corrosion of metals, where the surfaces of metals are degraded into more chemically stable oxides. However, the term ‘rusting’ is generally used to refer to the corrosion of objects made of iron or iron-alloys.
What is the Chemistry Behind the Rusting of Iron?
The exposure of iron (or an alloy of iron) to oxygen in the presence of moisture leads to the formation of rust. This reaction is not instantaneous, it generally proceeds over a considerably large time frame. The oxygen atoms bond with iron atoms, resulting in the formation of iron oxides. This weakens the bonds between the iron atoms in the object/structure.
The reaction of the rusting of iron involves an increase in the oxidation state of iron, accompanied by a loss of electrons. Rust is mostly made up of two different oxides of iron that vary in the oxidation state of the iron atom. These oxides are:
- Iron(II) oxide or ferrous oxide. The oxidation state of iron in this compound is +2 and its chemical formula is FeO.
- Iron(III) oxide or ferric oxide, where the iron atom exhibits an oxidation state of +3. The chemical formula of this compound is Fe2O3.
Oxygen is a very good oxidizing agent whereas iron is a reducing agent. Therefore, the iron atom readily gives up electrons when exposed to oxygen. The chemical reaction is given by:
Fe → Fe2+ + 2e–
The oxidation state of iron is further increased by the oxygen atom when water is present.
4Fe2+ + O2 → 4Fe3+ + 2O2-
Now, the following acid-base reactions occur between the iron cations and the water molecules.
Fe2+ + 2H2O ⇌ Fe(OH)2 + 2H+
Fe3+ + 3H2O ⇌ Fe(OH)3 + 3H+
The hydroxides of iron are also formed from the direct reaction between the iron cations and hydroxide ions.
O2 + H2O + 4e– → 4OH–
Fe2+ + 2OH– → Fe(OH)2
Fe3+ + 3OH– → Fe(OH)3
The resulting hydroxides of iron now undergo dehydration to yield the iron oxides that constitute rust. This process involves many chemical reactions, some of which are listed below.
- Fe(OH)2 ⇌ FeO + H2O
- 4Fe(OH)2 + O2 + xH2O → 2Fe2O3.(x+4)H2O
- Fe(OH)3 ⇌ FeO(OH) + H2O
- 2FeO(OH) ⇌ Fe2O3 + H2O
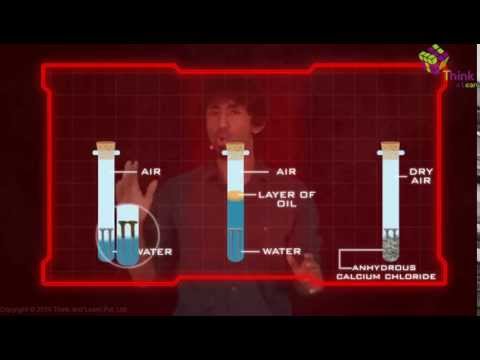
One similarity between all the chemical reactions listed above is that all of them are dependent on the presence of water and oxygen. Therefore, the rusting of iron can be controlled by limiting the amount of oxygen and water surrounding the metal.
Why is Rusting an Undesirable Phenomenon?
Rusting causes iron to become flaky and weak, degrading its strength, appearance and permeability. Rusted iron does not hold the desirable properties of iron. The rusting of iron can lead to damage to automobiles, railings, grills, and many other iron structures.
The collapse of the Silver Bridge in 1967 and the Mianus River bridge in 1983 is attributed to the corrosion of the steel/iron components of the bridge. Many buildings made up of reinforced concrete also undergo structural failures over long periods of time due to rusting.
Rusted iron can be a breeding ground for bacteria that cause tetanus. Cuts from these objects that pierce the skin can be dangerous.
Since rusting occurs at an accelerated rate in humid conditions, the insides of water pipes and tanks are susceptible to it. This causes the pipes to carry brown or black water containing an unsafe amount of iron oxides.
Factors that Affect the Rusting of Iron
Many factors speed up the rusting of iron, such as the moisture content in the environment and the pH of the surrounding area. Some of these factors are listed below.
- Moisture: The corrosion of iron is limited to the availability of water in the environment. Exposure to rains is the most common reason for rusting.
- Acid: if the pH of the environment surrounding the metal is low, the rusting process is quickened. The rusting of iron speeds up when it is exposed to acid rains. Higher pH inhibits the corrosion of iron.
- Salt: Iron tends to rust faster in the sea, due to the presence of various salts. Saltwater contains many ions that speed up the rusting process via electrochemical reactions.
- Impurity: Pure iron tends to rust more slowly when compared to iron containing a mixture of metals.
The size of the iron object can also affect the speed of the rusting process. For example, a large iron object is likely to have small deficiencies as a result of the smelting process. These deficiencies are a platform for attacks on the metal from the environment.
How can Rusting be Prevented?
Iron and its alloys are widely used in the construction of many structures and in many machines and objects. Therefore, the prevention of the corrosion of iron is very important. Some preventive methods are listed below.
Alloys that are Resistant to Rusting
Some alloys of iron are rust-resistant. Examples include stainless steel (which features a layer of chromium(III) oxide) and weathering steel.
COR-TEN steel rusts at a relatively slower rate when compared to normal steel. In this alloy, the rust forms a protective layer on the surface of the alloy, preventing further corrosion.
Galvanization
- Galvanization is the process of applying a protective layer of zinc on a metal. It is a very common method of preventing the rusting of iron.
- This can be done by dipping the metal to be protected in hot, molten zinc or by the process of electroplating.
- Zinc is a relatively cheap metal that sticks to steel easily. It also offers cathodic protection to the iron surface by acting as an anode. The zinc layer is corroded instead of the iron due to this.
- The disadvantages of galvanization are that it only provides protection from corrosion for a limited amount of time since the zinc layer is eaten up in the process. It is not very effective in highly corrosive areas (where cadmium coating can be used instead).
Cathodic Protection
- Providing the metals with an electric charge can help inhibit the electrochemical reactions that lead to rusting.
- This can be done by making the iron/steel a cathode by attaching a sacrificial anode to it.
- This sacrificial anode must have an electrode potential that is more negative than that of iron.
- Metals that are commonly used as sacrificial anodes are magnesium, zinc, and aluminium. Once they are corroded away, they must be replaced in order to protect the iron/steel.
Coatings
Many types of coatings can be applied to the surface of the exposed metal in order to prevent corrosion. Common examples of coatings that prevent corrosion include paints, wax tapes, and varnish.
Smaller objects are coated with water-displacing oils that prevent the rusting of the object. Many industrial machines and tools made of iron are coated with a layer of grease, which lubricates the metal to reduce friction and prevents rusting at the same time.
To learn more about the rusting of iron and other related concepts, such as the corrosion of metals, register with BYJU’S and download the mobile application on your smartphone.
Frequently Asked Questions – FAQs
What are physical and chemical changes?
A chemical transition is the result of a chemical reaction, and a physical change occurs where the structure of matter changes but not the chemical identity. Examples of chemical transformations include fire, frying, rusting, and rotting. Examples of physical changes are to simmer and freeze.
What defines a chemical change?
Chemical reactions requiring the rearrangement of atoms of one or more compounds and the modification of their chemical properties or structure resulting in the creation of at least one new substance: iron rust is a chemical alteration.
Is Melting zinc a chemical change?
A chemical reaction is a mechanism that happens by converting one or more compounds into one or more other compounds. No chemical reaction is registered. However, if the mixture absorbs energy in the form of heat, the zinc may react chemically with the sulphur to form the compound zinc sulphide (ZnS).
Which process is a chemical change?
Material modifications arise as a substance becomes a new material, called chemical synthesis or, similarly, chemical decomposition into two or three distinct compounds, combined with another. These mechanisms are called chemical reactions, and they are usually not reversible or by additional chemical reactions.
What is the importance of chemical change?
Chemical processes allow one to understand matter’s properties. We can learn its chemical properties by observing the way a sample interacts with another matter. These properties may be used to classify an unknown specimen or to predict how different kinds of matter may react with each other.

Thank you
Very good
Wow wonderful
very helpful
what is iron
Iron is a mineral, and its main purpose is to carry oxygen in the hemoglobin of red blood cells throughout the body so cells can produce energy.
What is the definition of Corrosion ?
Corrosion is when a refined metal is naturally converted to a more stable form such as its oxide, hydroxide or sulphide state this leads to deterioration of the material.
What is meant by unbalanced chemical equation ?
An unbalanced chemical equation lists the reactants and products in a chemical reaction but doesn’t state the amounts required to satisfy the conservation of mass.