NCERT Solutions for Class 10 Science Chapter 1 – CBSE Download Free PDF

NCERT Solutions for Class 10 Science Chapter 1 Chemical Reactions and Equations are outstanding study resources needed for the students studying in CBSE of Class 10. These NCERT Solutions are crafted as per the latest CBSE Syllabus by subject experts at BYJU’S. It is very important for the students to solve and study with the help of NCERT Solutions to get knowledge of the type of questions asked from the chapter, chemical reactions and equations.
Download Exclusively Curated Chapter Notes for Class 10 Science Chapter – 1 Chemical Reactions and Equations
Download Most Important Questions for Class 10 Science Chapter – 1 Chemical Reactions and Equations
Chapter 1 of NCERT Solutions for Class 10 Science mainly deals with writing chemical equations and writing and balancing chemical equations. Also, in this chapter, students learn the basics of chemical reactions, their types and the effects of a chemical oxidation reaction in our daily lives. The NCERT Solutions Class 10 Science Chapter 1 provides an overview of the main concepts in the chapter and help them to get well-versed in important topics such as writing chemical equations and balancing them.
The NCERT Solutions for Class 10 Science Chapter 1 have good weightage, and there will be around 4 questions asked from this chapter every year. The types of questions that appear in this chapter are mostly practice-based questions. Thorough knowledge and good practice will help learners score full marks on the questions asked in this chapter.
NCERT Solutions for Class 10 Science Chapter
Access answers to NCERT Class 10 Science Chapter 1 – Chemical Reactions and Equations
In-text questions set 1 Page number – 6
1. Why should a magnesium ribbon be cleaned before burning in the air?
Solution:
Magnesium ribbon should be cleaned before burning in air because Magnesium metal reacts with the atmospheric oxygen and forms Magnesium Oxide (MgO) layer which is a very stable compound. In order to prevent further reactions with Oxygen, it is therefore necessary to clean the ribbon by to remove the layer of MgO.
2. Write a balanced equation for the following chemical reactions.
i) Hydrogen + Chloride —-> Hydrogen chloride
ii) Barium chloride + Aluminium sulphate —-> Barium sulphate + Aluminium chloride
iii) Sodium + Water —-> Sodium hydroxide + Hydrogen
Solution:
i) H2 + Cl2 → 2HCl
ii) 3BaCl2 + Al2(SO4)3 →3BaSO4 + 2AlCl3
iii) 2Na + 2H2O → 2NaOH + H2
3. Write a balanced chemical equation with state symbols for the following reactions
i) Solutions of Barium chloride and Sodium sulphate in water react to give insoluble Barium sulphate and solution of Sodium chloride.
ii) Sodium hydroxide solution in water reacts with the hydrochloric acid solution to produce Sodium chloride solution and water.
Solution:
i) BaCl2 + Na2SO4 → BaSO4 + 2NaCl
ii) NaOH + HCl → NaCl + H2O
In-text questions set 2 Page number – 10
1. A solution of a substance, ‘X,’ is used for whitewashing.
(i) Name the substance ‘X’ and write its formula.
(ii) Write the reaction of the substance ‘X’ named in (i) above with water.
Solution:
i) The substance ‘X’ which is used in whitewashing is quick lime or Calcium Oxide and its formula is CaO.
ii) CaO + H2O → Ca(OH)2
2. Why is the amount of gas collected in one of the test tubes in Activity 1.7 double the amount collected in the other? Name this gas.
Solution:
In activity 1.7, gas collected in one of the test tubes is double of the amount collected in the other because water gets hydrolysed to release H2 and O2 gas. Here, after electrolysis two molecules of Hydrogen and one molecule of oxygen gas are released; hence, the amount of Hydrogen collected would be double than that of oxygen.
In-text questions set 3 Page number – 13
1. Why does the colour of copper sulphate solution change when an iron nail is dipped in it?
Solution:
When an iron nail is dipped in the copper sulphate solution, iron displaces copper from the copper sulphate because iron is more reactive than copper. Therefore, the colour of the copper sulphate solution changes. The reaction is
Fe + CuSO4 → FeSO4 + Cu
2. Give an example of a double displacement reaction other than the one given in Activity 1.10.
Solution:
Reaction Between silver nitrate (AgNO3) and Sodium chloride (NaCl) is an example of a double displacement reaction. During the reaction, negative and positive ions trade positions as a result of the formation of white silver chloride precipitate. The chemical reaction is given below.
Ag+ + NO3– + Na+ + Cl– → AgCl + Na+ + NO3–
3. Identify the substances that are oxidised and that are reduced in the following equation.
i) 4Na(s) + O2(g) → 2Na2O(s)
ii) CuO(s) + H2(g) → Cu(s) + H2O(l)
Solution:
The Sodium (Na) in the first equation is getting oxidized with the addition of Oxygen (O2), and the Copper (Cu) in the second equation is reduced due to the addition of Hydrogen (H2).
Exercise Questions Page number – 14-16
1. Which of the statements about the reaction below are incorrect?
2PbO(s) + C(s) → 2Pb(s) + CO2(g)
(a) Lead is getting reduced
(b) Carbon Dioxide is getting oxidised
(c) Carbon is getting oxidised
(d) Lead oxide is getting reduced
(i) (a) and (b)
(ii) (a) and (c)
(iii) (a), (b) and (c)
(iv) all the above
Solution:
(i) (a) and (b)
Explanation: (a) Because Oxygen is being removed and (b) Because the removed oxygen from Lead is added to the elemental Carbon.

2. Fe2O3 + 2Al → Al2O3 + 2Fe
The above reaction is an example of a
- Combination reaction
- Double displacement reaction
- Decomposition reaction
- Displacement reaction
Solution:
The answer is 4. Displacement reaction.
Explanation: The Oxygen from the Ferrous oxide is getting displaced to the Aluminium metal to form Aluminium Oxide. In this reaction, Aluminum is a more reactive metal than Fe. Therefore, Al will displace Fe from its oxide. This type of chemical reaction, called displacement reaction, is in which one of the elements displaces another. Here, less reactive metal is displaced by more reactive metal. Since one-time displacement is occurring, it is called a single displacement reaction.

3. What happens when dilute hydrochloric acid is added to iron fillings? Tick the correct answer.
- Hydrogen gas and Iron chloride are produced.
- Chlorine gas and Iron hydroxide are produced.
- No reaction takes place.
- Iron salt and water are produced.
Solution:
- Hydrogen gas and Iron chloride are produced.
Explanation: The Chlorine from Hydrogen chloride is displaced by the Iron fillings to undergo the following reaction.
2HCl + Fe → FeCl2 + H2

4. What is a balanced chemical equation? Why should a chemical equation be balanced?
Solution:
A balanced equation is the one in which number of different atoms on both the reactant and product sides are equal. Balancing chemical equations is necessary for the reaction should obey The Law of Conservation of mass. Balancing the chemical equation has no defined method and is purely a trial-and-error attempt.

5. Translate the following statements into chemical equations and balance them.
(a) Hydrogen gas combines with nitrogen to form ammonia.
(b) Hydrogen sulphide gas burns in the air to give water and sulphur dioxide.
(c) Barium chloride reacts with aluminium sulphate to give Aluminium chloride and a precipitate of barium sulphate.
(d) Potassium metal reacts with water to give potassium hydroxide and Hydrogen gas.
Solution:
(a) Unbalanced: H2 + N2 → NH3
Balanced: 3H2 + N2 → 2NH3

(b) Unbalanced: H2S + O2 → H2O + SO2
Balanced: 2H2S + 3O2 → 2H2O + 2SO2

(c) Unbalanced:
BaCl2 + Al2(SO4)3 → AlCl3 + BaSO4
Balanced: 3BaCl2 + Al2(SO4)3 → 2AlCl3 + 3BaSO4

(d) Unbalanced: K + H2O → KOH + H2
Balanced: 2K + 2H2O → 2KOH + H2

6. Balance the following chemical equations.
(a) HNO3 + Ca(OH)2 → Ca(NO3)2 + H2O
(b) NaOH + H2SO4 → Na2SO4 + H2O
(c) NaCl + AgNO3 → AgCl + NaNO3
(d) BaCl2 + H2SO4 → BaSO4 + HCl
Solution:
(a) 2HNO3 + Ca(OH)2 → Ca(NO3)2 + 2H2O

(b) 2NaOH + H2SO4 → Na2SO4 + 2H2O

(c) NaCl + AgNO3 → AgCl + NaNO3

(d) BaCl2 + H2SO4 → BaSO4 + 2HCl

7. Write the balanced chemical equation for the following reactions.
Calcium hydroxide + Carbon dioxide —-> Calcium carbonate + Water
Zinc + Silver nitrate —-> Zinc nitrate + Silver
Aluminium + Copper chloride —-> Aluminium chloride + Copper
Barium chloride + Potassium sulphate —-> Barium sulphate + Potassium chloride
Solution:
2Ca(OH)2 + 2CO2 → 2CaCO3 + 2H2O

Zn + 2AgNO3 → Zn(NO3)2 + 2Ag

2Al + 3CuCl2 → 2AlCl3 + 3Cu

BaCl2 + K2SO4 → BaSO4 + 2KCl

8. Write a balanced chemical equation for the following and identify the type of reaction of each case.
KBr + BaI2 → KI + BaBr2
ZnCO3 → ZnO + CO2
H2 + Cl → HCl
Mg + HCl → MgCl2 + H2
Solution:
2KBr + BaI2 → 2KI + BaBr2 (Double Displacement Reaction)

ZnCO3 → ZnO + CO2 (Decomposition Reaction)

H2 + Cl → 2HCl (Combination Reaction)

Mg + 2HCl → MgCl2 + H2 (Displacement Reaction)

9. What is meant by exothermic and endothermic reactions? Give examples.
Solution:
An endothermic reaction occurs when energy is absorbed from the surroundings in the form of heat (For example, Photosynthesis, melting of ice, evaporation). Conversely, an exothermic reaction is one in which energy is released from the system into the surroundings (Examples: Explosions, concrete setting, nuclear fission and fusion).

10. Why is respiration considered to be an exothermic reaction?
Solution:
For the survival of life, we require energy. We obtain this energy from the food we eat. The food molecules, through the process of digestion, is broken down into simpler molecule like glucose. These substances come in contact with the Oxygen present in our body cells to form Carbon dioxide and water along with a certain amount of energy (Respiration process). Since the energy is in the form of heat (that maintains our body temperature), respiration is considered to be an exothermic reaction. The reaction taking place is:
C6H12O6 + 6O2 → 6CO2 + 6H2O + Energy

11. Why are decomposition reactions called the opposite of Combination reactions? Write equations for decomposition reactions.
Solution:
Combination reaction is said to be the reaction between two or more molecules to form a larger molecule; whereas the decomposition reaction is defined as the splitting of larger molecules into two or more smaller molecules. This essentially explains that the decomposition reaction is the opposite of the combination reaction.
In most cases, the decomposition reaction is endothermic since the heat from the surrounding or induced heat is used to break the bonds of the larger molecule. A few examples of decomposition reactions are
ZnCO3 → ZnO + CO2
CaCO3 + Energy → CaO + CO2
2HgO → 2Hg + O2

12. Write one equation each for decomposition reactions in which energy is supplied in the form of heat, light or electricity.
Solution:
(a) Thermal decomposition reaction (Thermolysis)
Decomposition of potassium chlorate: When heated strongly, potassium chlorate decomposes into potassium chloride and oxygen. This reaction is used for the preparation of oxygen.
2KClO3 + Heat → 2KCl + 3O2
(b) Electrolytic decomposition reaction (Electrolysis)
Decomposition of sodium chloride: On passing electricity through molten sodium chloride, it decomposes into sodium and chlorine.
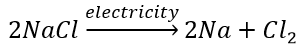
(c) Photodecomposition reaction (Photolysis)
Decomposition of hydrogen peroxide: In the presence of light, hydrogen peroxide decomposes into water and oxygen.
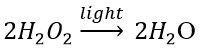

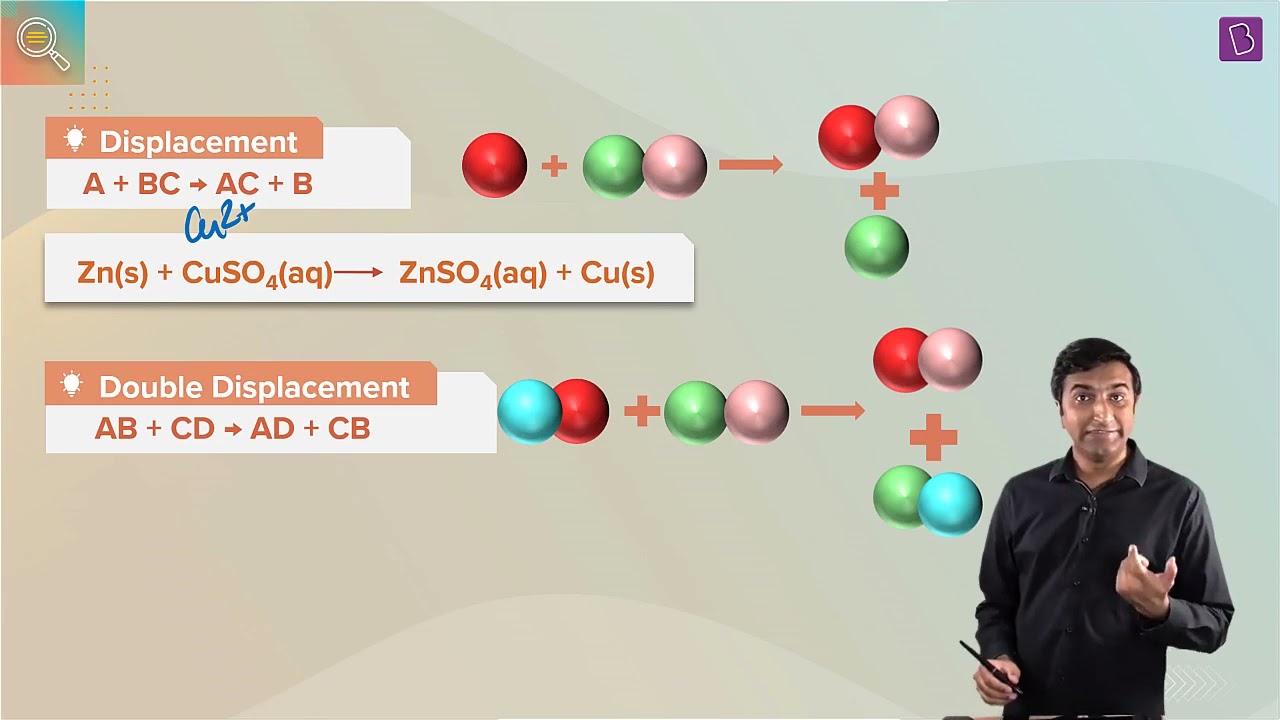
13. What is the difference between displacement and double displacement reactions? Write relevant equations for the above.
Solution:
A displacement reaction is one when a more reactive substance displaces a less reactive one from its salt solution, whereas a double displacement reaction is one where a mutual exchange of ions happens between two compounds.
In a displacement reaction, only a single displacement takes place, whereas in the double displacement reaction, as the name suggests, two displacement takes place between the molecules.
Example:
Displacement reaction
Mg + 2HCl → MgCl2 + H2
Double displacement reaction
2KBr + BaI2 → 2KI + BaBr2
14. In the refining of Silver, the recovery of silver from Silver nitrate solution involves displacement reaction by Copper metal. Write down the reaction involved.
Solution:
Cu(s) + 2AgNO3(aq) → Cu(NO3)2(aq) + 2Ag(s)

15. What do you mean by a precipitation reaction? Explain by giving examples.
Solution:
When two solutions containing soluble salts are combined, a double displacement reaction takes place in which the ions are exchanged between the compounds. When one of such compounds formed is in solid form (that is insoluble in aqua), then it settles down at the bottom of the container. This solid is known as the precipitate and the respective reaction is termed as the precipitation reaction. A few examples of precipitation reactions are
CdSO4(aq) + K2S(aq) → CdS(s) + K2SO4(aq)
2NaOH(aq) + MgCl2(aq) → 2NaCl(aq) + Mg(OH)2(s)

16. Explain the following in terms of the gain of oxygen with two examples each.
(a) Oxidation
(b) Reduction
Solution:
(a) In a chemical reaction, when the oxygen is added to the element to form its respective oxide it is the element being oxidised. Example:
4Na(s) + O2(g) → 2Na2O(s)
H2S + O2 → H2O + SO2

(b) In a chemical reaction, when the oxygen is removed from the compound, then it is said to be reduced. Example:
CuO(s) + H2(g) → Cu(s) + H2O(l)
2HgO → 2Hg + O2

17. A shiny brown coloured element ‘X’ on heating in the air becomes black in colour. Name the element ‘X’ and the black-coloured compound formed.
Solution:
The shiny brown-coloured element is Copper metal (Cu). When the metal is heated in air, it reacts with atmospheric oxygen to form copper oxide. Hence, the black-coloured compound is copper oxide.
2Cu(s) + O2(g) → 2CuO(s)

18) Why do we apply paint on iron articles?
Solution:
Iron articles are painted to prevent them from rusting. When left unpainted, the metal surface comes in contact with the atmospheric oxygen and in the presence of moisture it from Iron(III) oxide. But if painted, the surface does not come in contact with moisture and air, thus prevents Rusting.

19) Oil and Fat containing food items are flushed with Nitrogen. Why?
Solution:
The main purpose of flushing Nitrogen into food packets that contain oil and fat items is to prevent Rancidity which occurs when the oil or fat reacts with the oxygen letting out an unpleasant smell and taste. Therefore, by flushing Nitrogen, an unreactive surrounding is created, thus preventing rancidity.

20) Explain the following terms with one example each.
(a) Corrosion
(b) Rancidity
Solution:
(a) Corrosion is a process where a refined metal is oxidised by atmospheric oxygen to form a more stable compound, such as oxides. The metal gradually degrades during the corrosion process. Rusting of iron is a good example of corrosion where the iron is converted to Iron oxide. Millions of dollars are spent annually in preventing rusting from bridges and other monuments.

(b) The condition produced by the aerial oxidation of the oil and fat present in the food material that produces an unpleasant taste and smell. The rancidity is retarded when the food is kept inside the refrigerator since the low temperature does not promote the oxidation reaction.

| Also Access |
| NCERT Exemplar Solutions for Class 10 Science Chapter 1 |
| CBSE Notes for Class 10 Science Chapter 1 |
Topics Covered under Class 10 Chapter 1 Science Chemical Reactions and Equations
| Section Number | Topic |
| 1.1 | Chemical Equations |
| 1.1.1 | Writing a Chemical Equation |
| 1.1.2 | Balanced Chemical Equations |
| 1.2 | Types of Chemical Reactions |
| 1.2.1 | Combination Reaction |
| 1.2.2 | Decomposition Reaction |
| 1.2.3 | Displacement Reaction |
| 1.2.4 | Double Displacement Reaction |
| 1.3 | Have You Observed the Effects of Oxidation Reactions in Everyday Life? |
| 1.3.1 | Corrosion |
| 1.3.2 | Rancidity |
NCERT Solutions for Class 10 Science Chapter 1 Chemical Reactions and Equations
Chemical Reactions and equations is a very fundamental chapter that deals with basic knowledge of chemical reactions and equations. Students are advised to get tuned in with this chapter to avoid any sort of difficulty in understanding advanced topics of chemistry. If you are planning to take science in your higher secondary school, this is one of the important chapters you have to memorise for a very long period of time.
Features of NCERT Solutions for Class 10 Chapter 1 Science Chemical Reactions and Equations
- Students can get comprehensive practice in balancing different kinds of equations.
- NCERT Solutions will help students write different chemical equations appropriately
- Help them get a thorough practice of solving questions of varied difficulty before facing the CBSE examination.
A chemical reaction is an important phenomenon in our lives. We come across various chemical reactions like rusting of iron, curdling of milk, respiration, digestion and growth. In order to score good marks in the CBSE Class 10 examination, students must practise the NCERT Solution for Class 10 Science Chapter 1 provided here.
At BYJU’S, students get free NCERT Solutions for all the classes, subjects and chapters. To help them with CBSE exam preparation, BYJU’S also provides NCERT notes, videos, animations, previous years’ question papers, sample papers, NCERT exemplar solutions and textbooks.
To get access to all the resource material provided by us, students should register with BYJU’S site or download BYJU’S – The Learning App.
Frequently Asked Questions on NCERT Solutions for Class 10 Science Chapter 1
List out the topics and subtopics covered in the NCERT Solutions for Class 10 Science Chapter 1.
1.1 – Chemical Equations
1.1.1 – Writing a Chemical Equation
1.1.2 – Balanced Chemical Equations
1.2 – Types of Chemical Reactions
1.2.1 – Combination Reaction
1.2.2 – Decomposition Reaction
1.2.3 – Displacement Reaction
1.2.4 – Double Displacement Reaction
1.3 – Have you observed the effects of oxidation reactions in everyday life?
1.3.1 – Corrosion
1.3.2 – Rancidity
What are the key features of the NCERT Solutions for Class 10 Science Chapter 1?
1. Students will get enough practice in balancing different kinds of equations.
2. The solutions help students to write chemical equations effortlessly.
3. Provides a lot of practice questions of different difficulty levels to face the board exams without fear.
4. Each and every question is answered in a comprehensive way to help students with their CBSE exam preparation.









Good animation for explation
Good