1
Question
Which of the following statement is correct?
Which of the following statement is correct?
Open in App
Solution
The correct option is C (SCN)2 is pseudohalogen
An axion which is not a halogen but has similar property like halogen is called a pseudohalogen.A pseudohalogen is composed of two electronegative chemicals linked to each other like AB - BA.Hence 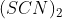 is a pseudohalogen.
is a pseudohalogen.
An axion which is not a halogen but has similar property like halogen is called a pseudohalogen.
A pseudohalogen is composed of two electronegative chemicals linked to each other like AB - BA.
Hence 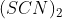 is a pseudohalogen.
is a pseudohalogen.
Suggest Corrections
0
View More
Join BYJU'S Learning Program
Join BYJU'S Learning Program
