Nitrogen cycle:
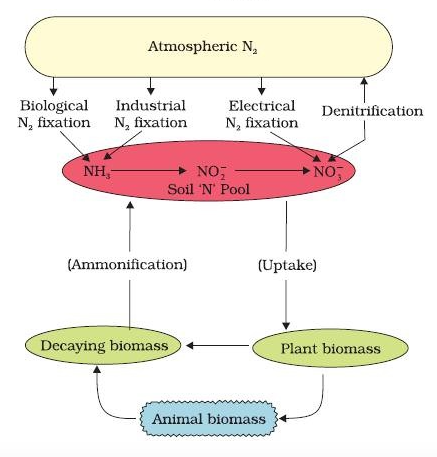
- The process of conversion of nitrogen (N2 ) to ammonia is termed nitrogen fixation.
- In nature, lightning and ultraviolet radiation provide enough energy to convert nitrogen to nitrogen oxides (NO, NO2, N2O).
- Industrial combustions, forest fires, automobile exhausts and power-generating stations are also sources of atmospheric nitrogen oxides.
- The decomposition of organic nitrogen of dead plants and animals into ammonia is called ammonification.
- Some of this ammonia volatilises and re-enters the atmosphere but most of it is converted into nitrate by soil bacteria in the following steps:
- Ammonia is first oxidised to nitrite by the bacteria Nitrosomonas and/or Nitrococcus. The nitrite is further oxidised to nitrate with the help of the bacterium Nitrobacter. These steps are called nitrification.
- The nitrate thus formed is absorbed by plants and is transported to the leaves. In leaves, it is reduced to form ammonia that finally forms the amine group of amino acids.
- Nitrate present in the soil is also reduced to nitrogen by the process of denitrification. Denitrification is carried by bacteria Pseudomonas and Thiobacillus.
Further Reading:
|
Related Links |
|
.
Comments