Aluminum Phosphate formula, also known as Aluminium monophosphate formula or Monoaluminum phosphate formula is explained in this article. Aluminum Phosphate is formed by the anion PO43- (phosphate) and cation Al3+ (aluminium). The molecular or chemical formula of Aluminum Phosphate is AlPO4.
Aluminum Phosphate occurs as a white crystalline powder. In its aqueous form, it is a colourless liquid. It is insoluble in water. Naturally, it occurs as a mineral berlinite. Aluminium monophosphate in its dihydrate form is found in minerals such as meta-variscite and variscite. Due to its piezoelectric material property, it is widely used in electrical and electronic industries.
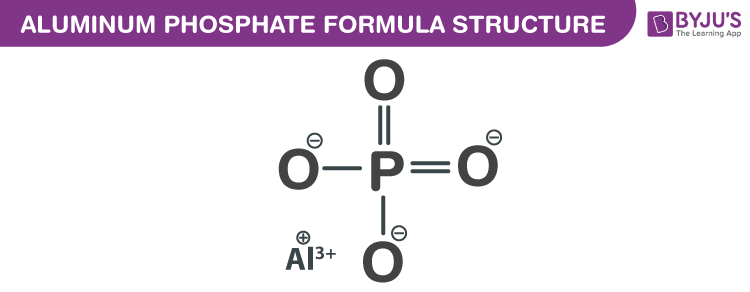
Aluminum Phosphate Formula Structure
Properties Of Aluminum Phosphate Formula
| Chemical formula | AlPO4 |
| Molecular weight | 121.9529 g/mol |
| Density | 2.566 g/cm3 |
| Boiling point | Decomposes |
| Melting point | 1,800 °C |
Monoaluminum phosphate is very dangerous to health. In high concentrations, it irritates eyes, mucosa, and skin. It can also harm the respiratory system. It is inflammable.
To learn more about Aluminum Phosphate formula from the expert faculties at BYJU’S, register now!
Comments