West Bengal Madhyamik Board 10th Standard Physical Science Exam Question Paper 2018- Free Download
The West Bengal Board Class 10 Physical Science 2018 Question Paper is considered as the best resource for those students who are preparing for the Class 10 board exams. The previous year papers set a proper foundation for the questions that will be asked in the Physical Sciences exam. Students can solve and practise the West Bengal Board Class 10 Physical Science 2018 Question Paper to ace the exams.
Students can click on the respective links and download the unsolved question paper PDF. Students can just click on the interactive PDF link and get the printable format of the question paper, as well. The questions and answers are also published online on the webpage.
Students are urged to familiarise themselves with the chief topics that are covered in the West Bengal Board Class 10 Physical Science textbook. The below given question paper is the perfect resource for students who want to get an overview of the question paper design of the WBBSE Class 10 Physical Science exam. Students are also advised to practise these previous year papers of West Bengal Board Class 10 Physical Science in order to revise the subject well. Referring to these questions will help the students to self analyse their performance and preparation level for the board exams. The students can also solve these papers regularly to know how to answer the questions effectively.
Download West Bengal Madhyamik Board 2018 Physical Science Question Paper
West Bengal Madhyamik Board Class 10 Physical Science 2018 Question
GROUP-A
- MCQ type questions. Four options are given as answers to all the questions given. Select and write the correct answer. [1X 15= 15]
1.1 Which of the following greenhouse gases has the maximum contribution towards global warming?
- N2O
- CH4
- CO2
- H2O Vapour
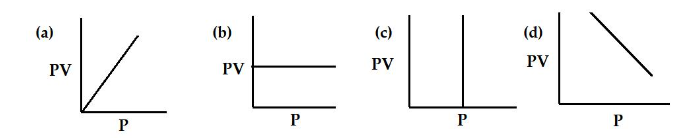
1.2 According to Boyle’s Law, which is PV-P Graph?
1.3 If the vapour density of a carbon containing gaseous substance is 13, which of the following is its molecular formula?
- CO2
- C2H4
- C2H6
- C2H2
.,
1.4 What is the unit of coefficient of linear expansion of a solid?
(a) m
(b) m-1
(c) °C-1
(d) °C
1.5 An object is placed between the optical centre and focus of a thin convex lens. What is the nature of the image of the object?
- real and inverted
- virtual and inverted
- real and erect
- virtual and erect
1.6 When a ray of light is incident perpendicularly on a transparent glass slab, what will be its angle of deviation?
- 0°
- 180°
- 30°
- 90°
1.7 Which of the units given below is the SI unit of resistance?
- volt
- ampere
- coulomb
- ohm
1.8 In domestic electric circuit the fuse wire is connected to which of the following?
(a) earth line
(b) live line
(c) neutral line
(d) both live and neutral
1.9 Which is the β-ray emitted from a radioactive element?
(a) a stream of electrons
(b) a stream of protons
(c) a stream of neutrons
(d) electromagnetic wave
1.10 How many groups are there in the long periodic table?
(a) 7
(b) 8
(c) 9
(d) 18
1.11 In formation of which of the following compounds, the octet rule is obeyed?
(a) NaCl
(b) LiH
(c) KCl
(d) CaO
1.12 Which of the following can conduct electricity?
(a) molten NaCl
(b) Liquid NaCl
(c) Solid NaCl
(d) aqueous solution of glucose
1.13 What will be the colour of the resulting solution when excess aqueous ammonia is added to an aqueous solution of copper sulphate?
(a) yellow
(b) green
(c) deep blue
(d) brown
1.14 In which of the following alloys is zinc present?
(a) bell metal
(b) brass
(c) bronze
(d) duralumin
1.15 Which of the following is a saturated hydrocarbon?
(a) C3H6
(b) C2H4
(c) C2H2
(d) C2H6
GROUP-B
2. Answer the following questions (options are given for some questions)
2.1 Mention one use of biogas
Or
What is the role of NO in decomposition of ozone in the ozone layer?
2.2 Among charcoal, petrol and ethanol, which one is a fossil fuel?
2.3 Under constant pressure, at what temperature in a degree Celsius, the volume of an ideal gas will be zero according to Charles Law?
2.4 What is the unit of M in the equation PV= (W/M) RT?
(symbols have usual meaning)
2.5 Whether the following statement is ‘true’ or ‘false’
The real expansion of any liquid depends on the expansion of the vessel in which it is kept.
Or
Among iron, inver and popper, which one has the least Coefficient of linear expansion?
2.6 Between the angle of incidence and the angle of refraction, which one is greater when light travels from a rarer to a denser medium?
2.7 What type of mirror is used in the viewfinder of a motor car?
2.8 How does the resistance of semiconductor change with increase of temperature?
2.9 What type of energy is transformed to electrical energy in a dynamo?
2.10 Arrange α , β, γ-rays in ascending order of their penetrating power.
Or
Which kind of nuclear reaction is the source of the sun’s energy?
2.11 Match the right column with the left column [1×4]
| Left Column | Right Column |
| 2.11.1 An Alkali metal |
|
| 2.11.2 An element whose anion accelerates rusting of iron |
|
| 2.11.3 Extracted from Haematite | (c) K |
| 2.11.4 Most electronegative elements | (d) Cl |
2.12 What type of chemical bond is present in CaO?
2.13 What is used as a cathode to electroplate silver over a copper spoon?
Or
Give an example of a compound whose aqueous solution is a weak electrolyte.
2.14 During electrolysis which which electrode is called a cathode?
2.15 Give one use of liquid Ammonia.
Or
Write the formula of the precipitate formed when aqueous ammonia solution is added to aqueous solution of aluminium chloride.
2.16 In the laboratory preparation of nitrogen aqueous solutions, which compound is mixed with aqueous solution of ammonium chloride and heated?
2.17 What is the IUPAC name of CH3CH2CHO?
Or
Write the structural formula of the positional isomer of CH3CH2CH2OH.
2.18 Mention one use of tetrafluoroethylene
GROUP-C
3. Answer the following questions (Alternatives are given for some questions) [2×9=18]
3.1 What is methane hydrate?
3.2 The pressure of a fixed mass of a gas at a temperature of 0 °C is doubled while the volume is halved. What will be the final temperature of the gas?
Or
Under constant pressure, a fixed mass of a gas is heated from 0 °C to 546 °C. What is the ratio of the final volume of the gas with its initial volume?
3.3 What is meant by the optical centre of a convex lens?
Or
Why does the earth’s sky appear blue during daytime?
3.4 State Lenz’s law related to electromagnetic induction.
3.5 Write with an example, how according to Lewis concept, a covalent bond is formed.
Or
Why the bond in sodium chloride cannot be expressed as Na-Cl?
3.6 Give one example each of a liquid and a solid covalent bond.
3.7 Write the balanced chemical equation of what happens when H2S gas is passed through an aqueous copper sulphate solution.
3.8 Write down the cathode reaction when an aqueous solution of MSO4(M=metal) is electrolysed. Write with reason whether the reaction is oxidation or reduction.
Or
Give one use each of copper and aluminium.
3.9 What is the condition of substitution reaction of methane with chloride? Write the balanced chemical equation for the first step of the reaction.
Or
Write with a balanced chemical equation what happens when ethanol reacts with metallic sodium.
GROUP-D
4. Answer the given questions(Make a note of the alternative questions)
4.1 Establish the ideal gas equation on the basis of Boyle’s law, Charles law and Avogadro’s law.
4.2 SO2 required for industrial production of sulphuric acid is produced by burning iron pyrites in excess air current. The chemical equation of the current is given below:
4FeS2 + 11O2 → 2Fe2O3 + 8SO2
How many grams of FeS2 is required for the production of 512g of SO2?
[Fe=56, S=32, O=16]
Or
By heating 200 g of metal carbonate, 112 g of metal oxide on a gaseous compound are produced. Vapour density of the gaseous compound is 22. How many moles of gaseous compound is produced in the reaction?
4.3 What is thermal conductivity? What is its SI unit?
4.4 How can an erect and magnified image be formed with the help of a convex lens? With the help of which type of lens, long-sightedness can be rectified?
4.5 If the velocity of light in a medium is 2 x 108 m/s , what will be the refractive index of that medium?
Or
The refractive index of a medium with respect to the air is √2. If the angle of incidence of a ray of light in air is 45° determine the angle of deviation for that ray in case of refraction.
4.6 Write joule’s law related to the heating effect of the current.
4.7 Calculate the equivalent resistance when a wire of 10 ohm is divided into two equal parts and connected in parallel combination.
Or
There are 60 watt lamp and two 80 watt fans in a house. The lamp and the fan runs for 5 hours daily. Find out the expense in a month if a unit of electricity cost Rs, 4/- (assume that one month= 30 days).
4.8 Compare the charge and ionising power of α and γ rays. Mention one use of radioactivity.
4.9 What is meant by ionisation energy of an atom of elements? Arrange Li, Rb, K and Na in the increasing order of their ionisation energy.
Or
Mention similarity of properties of hydrogen with one property of Group I elements and two properties of Group 17 elements.
4.10 What are present along with pure alumina in the molten mixture which is electrolysed for the extraction of aluminium by electrolysis? What is used as cathode and anode in this electrolysis?
4.11 Write the condition and balanced chemical equation for the industrial production of ammonia by Haber’s process.
4.12 The molecular formula of the organic compound is C2H4O2, the compound is soluble in water and on addition of NaHCO3 to the aqueous solution of the compound, CO2 is evolved. Identify the organic compound. Write with condition and balanced chemical equation, the reaction of the compound with ethanol.
Or
Compare three properties of the organic and the inorganic compound.
GROUP-E
[These questions are for external candidates only]
5. Answer any four from the following questions [1×4=4]
5.1 In which layer of the atmosphere does the storm occur?
5.2 The alkaline earth metals are placed in which group of the periodic table?
5.3 In what combination, two resistance is to be conducted so that the equivalent resistance is smaller than both the resistance?
5.4 Write the name of the gas that smells like rotten eggs.
5.5 What is the product of pressure and volume of 2g of hydrogen at STP?
[H=1]
6. Answer any three from the following questions. [2×3=6]
6.1. Give the name and formula of one ore of copper.
6.2 Why is a convex lens called convergent lens?
6.3 Write with balanced chemical equation of what happens when ethylene reacts with bromine.
6.4 How is the concept of resistance obtained from Ohm’s law?







Comments