The molecules of air frequently bump into the walls of the vessel. These impacts apply energy on the walls of the vessel which is not anything but termed as pressure. The pressure exerted by the gas is force per unit area. Though as we know, the air is a mixture of gases like nitrogen, oxygen, etc. and not a single compound. The pressure that is contributed by every gas is termed its partial pressure. Partial pressure can, therefore, be explained as the pressure applied if that gas by itself had engaged the volume. Therefore, the entire pressure of air will be the summation of partial pressures of oxygen, nitrogen, etc.
The aggregate pressure is, therefore, the summation of partial pressures of the ideal gases. This is articulated by Dalton’s law of partial pressures.
The aggregate pressure is, therefore, the summation of partial pressures of the ideal gases. This is articulated by Dalton’s law of partial pressures.
Ptotal=P1+P2+… +Pn
where Ptotal is the aggregate pressure of air, P1+P2+… +Pn are partial pressures of the ideal gases
Solved Examples
Problem 1: To a tank containing O2 at 1.0atm and N2 at 2.0atm, we add an anonymous quantity of CO2 up until the total pressure inside the tank is 4.6atm. What is CO2’s partial pressure?
Answer:
Dalton’s law states that the addition of CO2 does not upset the partial pressures of the O2 and N2 already existing in the tank. The partial pressure of O2 and N2 remain at 2.0atm, respectively, and their total is 3.0atm. The partial pressure of the added CO2 must be 1.6 atm if the new pressure is 4.6atm, Therefore, when the final pressure is 4.6atm, the partial pressures are
Ptotal=P1+P2+… +Pn
4.6atm =1.0atm+2.0atm+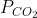
Comments