Electron Transport Chain is a series of compounds where it makes use of electrons from electron carrier to develop a chemical gradient. It could be used to power oxidative phosphorylation. The molecules present in the chain comprises enzymes that are protein complex or proteins, peptides and much more.
Large amounts of ATP could be produced through a highly efficient method termed oxidative phosphorylation. ATP is a fundamental unit of metabolic process. The electrons are transferred from electron donor to the electron acceptor leading to the production of ATP. It is one of the vital phases in the electron transport chain. Compared to any other part of cellular respiration the large amount of ATP is produced in this phase.
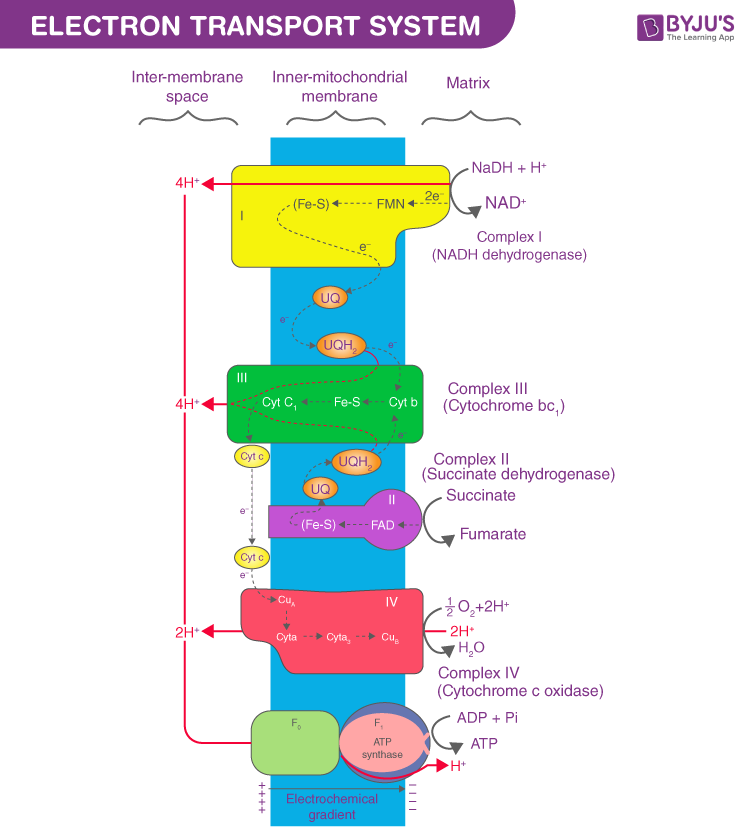
Electron transport is defined as a series of redox reaction that is similar to the relay race. It is a part of aerobic respiration. It is the only phase in glucose metabolism that makes use of atmospheric oxygen. When electrons are passed from one component to another until the end of the chain the electrons reduce molecular oxygen thus producing water. The requirement of oxygen in the final phase could be witnessed in the chemical reaction that involves the requirement of both oxygen and glucose.
Electron Transport Chain in Mitochondria
A complex could be defined as a structure that comprises a weak protein, molecule or atom that is weakly connected to a protein. The plasma membrane of prokaryotes comprises multi copies of the electron transport chain.
Complex 1- NADH-Q oxidoreductase: It comprises enzymes consisting of iron-sulfur and FMN. Here two electrons are carried out to the first complex aboard NADH. FMN is derived from vitamin B2.
Q and Complex 2- Succinate-Q reductase: FADH2 that is not passed through complex 1 is received directly from complex 2. The first and the second complexes are connected to a third complex through compound ubiquinone (Q). The Q molecule is soluble in water and moves freely in the hydrophobic core of the membrane. In this phase, an electron is delivered directly to the electron protein chain. The number of ATP obtained at this stage is directly proportional to the number of protons that are pumped across the inner membrane of the mitochondria.
Complex 3- Cytochrome c reductase: The third complex is comprised of Fe-S protein, Cytochrome b, and Cytochrome c proteins. Cytochrome proteins consist of the heme group. Complex 3 is responsible for pumping protons across the membrane. It also passes electrons to the cytochrome c where it is transported to the 4th complex of enzymes and proteins. Here, Q is the electron donor and Cytochrome C is the electron acceptor.
Complex 4- Cytochrome c oxidase: The 4th complex is comprised of cytochrome c, a and a3. There are two heme groups where each of them is present in cytochromes c and a3. The cytochromes are responsible for holding oxygen molecule between copper and iron until the oxygen content is reduced completely. In this phase, the reduced oxygen picks two hydrogen ions from the surrounding environment to make water.
This was a simple introduction to Electron Transport Chain. For more information, visit BYJU’S.

Comments