
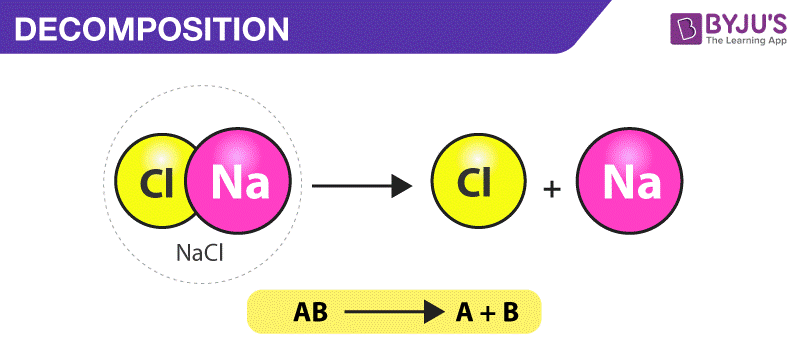
What is a Decomposition Reaction?
A decomposition reaction can be defined as a chemical reaction in which one reactant breaks down into two or more products.
Table of Contents
- Decomposition reaction examples
- Double Decomposition Reaction
- Uses of Decomposition Reactions
- Frequently Asked Questions – FAQs
Recommended Video
Decomposition Reactions

General Reaction Format
The general format of a decomposition reaction is provided below.
AB -> A + B
Where AB is the parent molecule (reactant) and A & B are the product molecules.
Decomposition reaction examples
Decomposition reactions happen all around us, but we often don’t notice them. Some common examples of decomposition reactions are provided below.
- The decomposition of carbonic acid in soft drinks, which can be represented by the chemical equation H2CO3 → H2O + CO2
- The electrolysis of water to yield hydrogen and oxygen.
What is the Opposite of ‘Decomposition Reaction’?
The opposite of a decomposition reaction is a combination reaction. Such reactions involve the formation of a single product from two or more reactants.
Are all Decomposition Reactions Endothermic?
No, not all decomposition reactions are endothermic. A decomposition reaction can be both endothermic or exothermic. However, the latter is more common than the former. For example:
- The decomposition of NO to N2 & O2 is exothermic.
- Decomposition of ozone (O3) to oxygen (O2) is exothermic.
What are the Applications of Decomposition Reactions?
One major application of decomposition reactions is in the extraction of metals from their ores. For example, zinc can be obtained from calamine by subjecting it to a decomposition reaction. In a similar manner, sodium can be obtained from sodium chloride (NaCl).
Double Decomposition Reaction
A double decomposition reaction is a type of decomposition reaction in which two constituent reactants interchange positive and negative ions and form two new compounds.
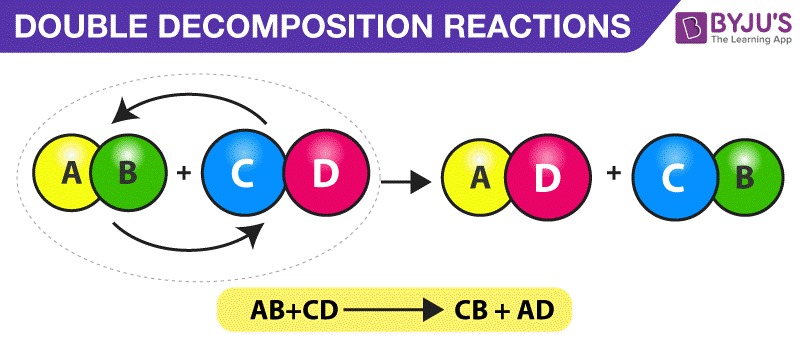
Example
HCl(aq) + NaOH(aq) → NaCl(aq) + HOH(l)
Types of Decomposition Reaction
The three primary types of decomposition reactions are:
Thermal decomposition reaction
A thermal decomposition reaction can be defined as a decomposition reaction which is activated by thermal energy. In other words, a thermal decomposition reaction requires energy to be supplied to the reactants in the form of heat. Such reactions are generally endothermic since energy is required to break the chemical bonds and separate the constituent elements. A common example of a thermal decomposition reaction is provided below.
CaCO3 → CaO + CO2
When heated, calcium carbonate decomposes into calcium oxide and carbon dioxide. This process is employed in the manufacturing of quick lime, which is an important substance in many industries.
Electrolytic decomposition reaction
An electrolytic decomposition reaction is a type of decomposition reaction in which the activation energy for decomposition is provided in the form of electrical energy. An example of an electrolytic decomposition reaction is the electrolysis of water, which can be represented by the following chemical equation: 2H2O→ 2H2 + O2
Photo decomposition/ photolytic decomposition/ photochemical decomposition
A photodecomposition reaction is a type of decomposition reaction in which the reactant is broken down to its constituents by absorbing energy from photons. An example of a photo decomposition reaction is the decomposition of ozone into dioxygen and an oxygen radical, as represented by the chemical equation provided below.
Uses of Decomposition Reactions
- Manufacture of cement or calcium oxide.
- For metallurgical processes: Extraction of metals from their oxides, chlorides, etc.
- The relief from acid indigestion.
- Thermite welding.
Frequently Asked Questions – FAQs
What is decomposition reaction?
A decomposition reaction can be defined as a chemical reaction in which one reactant breaks down into two or more products.
Are all decomposition reactions endothermic?
No, not all decomposition reactions are endothermic. A decomposition reaction can be both endothermic and exothermic. However, the latter is more common than the former.
What are the applications of decomposition reactions?
One major application of decomposition reactions is in the extraction of metals from their ores. For example, zinc can be obtained from calamine by subjecting it to a decomposition reaction. In a similar manner, sodium can be obtained from sodium chloride (NaCl).
What is a double decomposition reaction?
A double decomposition reaction is a type of decomposition reaction in which two constituent reactants interchange positive and negative ions and form two new compounds.
What is an electrolytic decomposition reaction?
An electrolytic decomposition reaction is a type of decomposition reaction in which the activation energy for decomposition is provided in the form of electrical energy. An example of an electrolytic decomposition reaction is the electrolysis of water, which can be represented by the following chemical equation.
2H2O → 2H2 + O2
To learn more about the different types of chemical reactions, register with BYJU’S.

thanks
Thank you
its very helpful
thank you
condition for decompoition reaction
Most decomposition reactions require energy either in the form of heat, light or electricity. Absorption of energy causes the breaking of the bonds present in the reacting substance which decomposes to give the product.