Table of Contents
What is an Organic Compound?
An organic compound is a class of chemical compounds in which one or more atoms of carbon are covalently bonded to other atoms. The Study of organic compounds is vast. Millions of compounds can be categorised under organic compounds. There are some organic compounds which consist of functional groups.
What is a functional group?
A functional group is an individual atom or a compound formed uniquely. A functional group is the most reactive site in an organic molecule. Compounds with a similar functional group have similar type of reactions.
For example, CH3OH and CH3CH2OH have similar type of reactions as they have the same functional group OH. These liberate hydrogen on reaction with sodium metal.
In an organic compound, firstly, the functional group is identified which gives us the appropriate suffix. Then the longest carbon chain having the functional group is chosen in such a manner that the functional group gets the lowest number in the chain.
In case of multiple functional groups in a compound, one of the functional groups is chosen on the basis of the priority list and is referred to as the principal functional group. The compound is named on the basis of that functional group. The remaining functional groups are known as the substituents and are named using the appropriate prefixes.
The priority list of the functional group can be given as:
-COOH > -SO3H > -COOR (R= alkyl group) > -COCl > -CONH2 > -CN > -HC=O > >C=O > -OH > -NH2 > >C=C< > -CC-
Functional groups like -R, C6H5-, halogens (F, Cl, Br, I), -NO2, alkoxy (-OR) etc., are always used as prefix substituents.
If more than one same functional group is present in the compound, then they are indicated as di, tri, … etc. before the suffix and full name of the parent alkane is written.
- Name the compound given below.
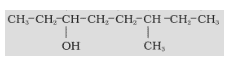
- In this compound, the functional group present is –OH.
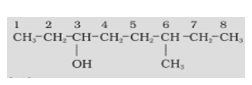
- The longest chain which contains the functional group has 8 carbon atoms. Hence, the saturated hydrocarbon is octane.
- The alcohol functional group is present on the 3rd position, and a methyl group is on the 6th position.
- Hence, the IUPAC name will be 6- Methyloctan-3-ol.
- Name the functional group given below:

This compound has the functional group as ketone (>C=O). Hence, the suffix will be one. And there are two ketone groups. So we will use di- before suffix as dione. Continuing in the same manner as above we get the name Hexane-2, 4-dione.
In this article, we read about the nomenclature of organic compounds having functional groups. To know more about the organic compound having benzene groups and isomerism, download BYJU’S- the learning app.

Comments