You would have observed changing states of matter when ice cubes melt from solid into liquid water or when water boils into vapor, but have you wondered why substances change form? Changing states of matter occur when matter loses or absorbs energy. When a substance absorbs energy; the atoms and molecules move more rapidly and this increased kinetic energy pushes particles far enough that they change form. This energy is usually heat or thermal energy. In this article, let us understand the science behind the changing states of matter.
| Table of Contents: |
What are Changes of State?
A change of state is a physical change in a matter. They are reversible changes and do not involve any changes in the chemical makeup of the matter. Common changes of the state include melting, freezing, sublimation, deposition, condensation, and vaporization. These changes are shown in the figure given below.
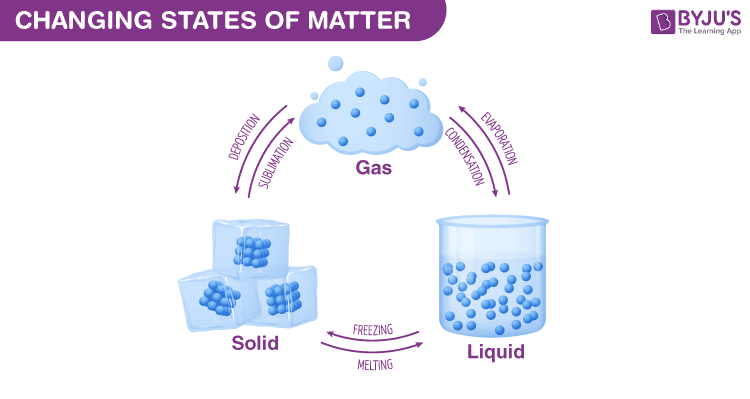
Why do Phase Changes Occur?
When temperature or pressure change of a system occurs, phase changes occur. When the temperature or pressure increases, the interaction between the molecules increases. Similarly, when the temperature decreases, it is easier for molecules and atoms to settle into a more rigid structure.
Changes Between Liquids and Solids
How would you make ice cubes in a tray? First, you would fill the tray with water from a tap. Then you would place the tray in the freezer compartment of a refrigerator. The freezer is very cold. What happens next?
Freezing
Heat transfer occurs between the warmer tray and the colder air in the freezer. The warm water loses heat to the cold air in the freezer. This heat transfer occurs until no energy is available for the particles to slide past each other. This forces them to remain in fixed positions, locked in place by the force of attraction between them. This way liquid water is changed into solid ice. The process of liquid water changing to solid ice is termed as freezing. The temperature at which it occurs is known as the freezing point.
Melting
If you took out the ice cubes from the freezer and placed them in a warm room, the ice would absorb energy from the warmer air around them. This absorbed energy would facilitate them to overcome the force of attraction holding them together, enabling them to slip out of the fixed position that they held as ice. The process in which a solids change to a liquid is called melting. The melting point is the temperature at which a solids change to a liquid.
You might want to read the following articles for a deeper understanding of the topic.
Watch the video below to clearly understand why water changes to solid when the temperature is reduced and to a gas when the temperature is increased?

Changes Between Liquids and Gases
If you fill a pot with cold tap water and heat it on a hot stovetop, the water heats up. Heat energy travels from the stovetop to the pot, and the water absorbs the energy from the pot. What happens to the water next?
Vaporization
If the water is hot enough, it starts to boil. Bubbles of water vapor are formed in the boiling water. This happens as particles of liquid water gain enough energy to completely overcome the force of attraction between them and change to the gaseous state. The bubbles rise through the water and escape from the pot as steam. The process in which a liquid boils and changes to a gas is called vaporization. The temperature at which a liquid boils is its boiling point.
Condensation
When you take a hot shower in a closed bathroom, the mirror is likely to fog up. You may wonder why does this happen? Some hot water from the shower evaporates and when it comes in contact with cooler surfaces such as the mirror, it cools and loses energy. The cooler water particles no longer have the energy to overcome the forces of attraction between them. They come together and form droplets of liquid water. This process in which a gas changes to liquid is known as condensation.
Changes Between Solids and Gases
Solids that change to gas pass through the liquid state first. However, sometimes solids change directly to gases and skip the liquid state. The reverse can also occur. Sometimes gases change directly to solids.
Sublimation
The process in which solids directly change to gases is known as sublimation. This occurs when solids absorb enough energy to completely overcome the forces of attraction between them. Dry ice is an example of solids that undergo sublimation.
Five Changes of State are:
- Melting
- Freezing
- Evaporation
- Condensation
- Sublimation
The process by which a substance changes from the solid phase to the liquid phase is known as melting.
The process by which a substance changes from the liquid phase to the solid phase is known as freezing.
The process by which a substance changes from the liquid phase to the gaseous phase is known as evaporation. The process by which a substance changes from the gaseous phase to the liquid phase is known as condensation. The transition of the solid phase to the gaseous phase without passing the intermediate liquid phase is known as sublimation.
Conclusion
It will interest you to know that every object in existence undergoes a state change. It is only a question of the amount of heat supplied to the substance. If you supply enough heat, everything on this planet can be made to change its state. The thing is though not every substance has to follow the solid-liquid-gas path. Some substances can naturally change from their solid-state to their gaseous state without entering the liquid state. This phenomenon is known as Sublimation. Examples of sublimation are, the element Iodine, Dry ice (solid CO2) and high-quality coal which at high-temperature burns and sublimates into vapour.
Frequently Asked Questions – FAQ’s
When solids reach their melting point, what do they become?
What is the boiling point?
What is the melting point?
What is the process in which solids directly transform into a gas?
What is evaporation?
The world around you is filled with interesting facts like these. Learn all about them at BYJU’S. Stay tuned to BYJU’S to learn more interesting concepts like changing states of matter with the help of engaging video lessons.

Nice information my teacher was so happy with my answer in particular. Thanks a lot. God bless😘😘😘😘❤️
very good for thank you