Most of us might have observed that whenever we dump household solid wastes at dumpsites, a part of the waste disappears after few days (biodegradable wastes) but some of the waste starts accumulating at the place (non-biodegradable wastes). The polymers which can decompose in a few days are decomposed by the action of microorganisms and are known as Biodegradable Polymers. The polymers which are not decomposed by the action of microorganisms and are referred to as non-biodegradable polymers.
To control these diseases and infections, we have started using eco-friendly synthetic polymers. This kind of polymers consists of similar functional groups as that of the biopolymers. They can be synthetically made by adding molecules of ester, anhydride, and amide. These molecules form a linkage between other molecules.
Table of Contents
What are Biodegradable Polymers?
These are those polymers which get decomposed under aerobic or anaerobic conditions, as a result of the action of microorganism/enzymes. The materials develop it like starch, cellulose, and polyesters. Aliphatic polyesters are the most commonly used polymers of this type.
Examples Of Biodegradable Polymers
Some examples are given as follows:-
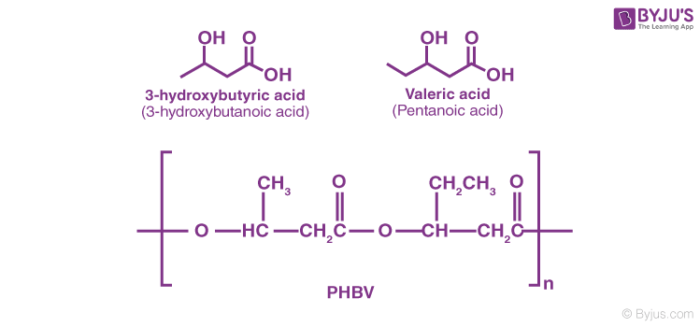
Poly β-hydroxybutyrate – co-β-hydroxy valerate (PHBV): It is derived by combining 3-hydroxy butanoic acid and 3-hydroxy pentanoic acid, in which monomers are cross-linked by an ester linkage. It decomposes to form carbon dioxide and water. It is brittle in nature, and it can be used in the production of drugs and the manufacturing of bottles.
Nylon 2–Nylon 6: It is a polyamide copolymerization of glycine (H2N−CH2−COOH) and aminocaproic acid (H2N−(CH2)5−COOH).
Polyhydroxybutyrate (PHB): It is formed by the condensation of hydroxybutyric acid (3-hydroxy butanoic acid) molecules.
What is Non-Biodegradable Polymer?
They consist of long chains of carbon and hydrogen atoms. These molecules form an interatomic type of bonding and is adamant meaning it is tough for microbes to break the bonds and digest them. Thus a long period is required to decompose them.
Examples Of Non-Biodegradable Polymer
Some examples are:
Polyethene: They are of three types:
-
-
-
- Linear high-density polyethene (HDPE)
- Branched low-density polyethene (LDPE)
- Ultra-high molecular weight polyethylene(UHMWPE)
-
-
These have high strength and lubricity and are also used in orthopaedic implants and catheters.
Poly (tetrafluoroethylene) (PTFE) (Teflon): They are chemically and biologically inert and have high lubricity. They are used in hollow fibres for enzyme immobilization, wiring in aerospace, etc.
Related Topics
Recommended Videos

So, now we know about the biodegradable polymer and non-biodegradable polymers and how they are made. Learn more about the polymers and their types by downloading BYJU’S learning app.

So are toothbrush’s that have nylon-6 for bristles biodegradable?
No, nylon-6 is not biodegradable.
Which is a not a non biodegradable?
– nylon 6,6
– nylon 6,10
– nylon 6
None
None of these polymers listed above are known to be biodegradable.