What is Barium bromide?
BaBr2 is an important chemical compound with the chemical name Barium bromide. It is also called Barium (2+) dibromide or Barium bromide anhydrous. It appears as a white solid which is soluble in water and is toxic in aqueous form.
Barium (2+) dibromide, crystallises to lead chloride motifs resulting in white, deliquescent, orthorhombic crystals. In its aqueous form, it acts like a simple salt. It reacts with the sulphate ion of sulpuric acid (H2SO4) to give barium sulphate (BaSO4) precipitates.
BaBr2(aq) + SO42− → BaSO4(s) + 2 Br−(aq)
Table of Contents
- Structure of Barium Bromide
- Properties of Barium Bromide
- Production of Barium Bromide
- Uses of Barium Bromide
- Health Hazards of Barium Bromide
- Frequently Asked Questions- FAQs
Structure of Barium bromide – BaBr2
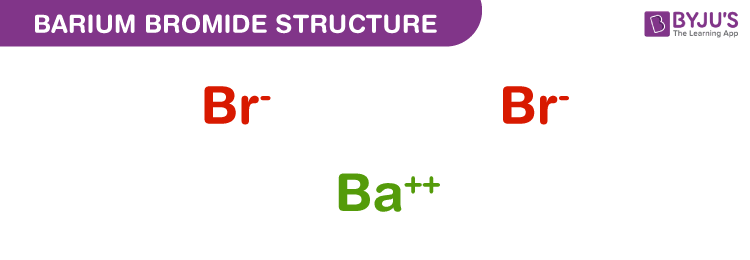
The exact mass of barium bromide is 297.74 g/mol and the monoisotopic mass is 297.742 g/mol. The number of hydrogen bond donors equals to zero whereas the number of hydrogen bond acceptors equals to 2. The compound is canonicalised and has three covalently bonded units.
Properties of Barium bromide – BaBr2
| BaBr2 | Barium bromide |
| Molecular weight of BaBr2 | 297.14 g/mol |
| Density of Barium bromide | 4.78 g/cm3 (anhydrous) |
| Boiling point of Barium bromide | 1,835 °C |
| Melting Point of Barium bromide | 857 °C |
Production of Barium bromide
BaBr2 can be prepared from barium carbonate or barium sulphide by reacting with hydrobromic acid (HBr) to produce hydrated barium bromide. The reaction is as follows:
BaS + 2HBr → BaBr2 + H2S
BaCO3 + 2HBr → BaBr2 + CO2 + H2O
Barium (2+) dibromide can be crystallised out from the solution in its dihydrate form. On heating at around 120 °C, BaBr2·2H2O gives the anhydrous form.
Uses of Barium bromide- BaBr2
- Barium bromide is used as a precursor in chemicals.
- Used in photography.
- It was used in a process called fractional crystallisation to purify radium.
- Used in the manufacturing of other bromides.
- Used in the production of phosphors.
Health hazards of Barium bromide- BaBr2
Barium bromide anhydrous along with a few other water-soluble barium salts, is considered toxic and when swallowed causes severe poisoning.
Learn more about the Structure, physical and chemical properties of BaBr2 from the experts at BYJU’S.
Frequently Asked Questions- FAQs
1. What is barium bromide used for?
Barium bromide was used in a process called fractional crystallisation to purify radium. Now it is used as a precursor in chemicals, Used in photography, and manufacturing of other bromides.
2. What kind of bond is barium bromide?
Barium bromide is an ionic compound. Barium loses two electrons to form Ba2+ cation and each bromine accepts one electron to form Br–.
3. Does barium bromide dissociate in water?
Barium bromide is a polar compound. It dissolves well in water and is toxic. It is dissociated by water to form Ba2+ and Br– ions.
4. How does barium bromide form?
Barium bromide is prepared by treating barium sulphide or barium carbonate with hydrobromic acid.
5. What state of matter is barium bromide?
Barium bromide appears as a white solid which is soluble in water.

Comments