What is a Coupling Reaction?
The term ‘coupling reaction’ refers to the class of organic reactions that involve the joining of two chemical species (usually with the help of a metal catalyst). An important type of coupling reaction is the reaction of an organic halide with an organometallic compound having the general formula R-M which facilitates the formation of a new carbon-carbon bond. If the organic halide in this reaction has the general formula R’-X, the compound formed as a product will have the formula R-R’.
An illustration of a coupling reaction is provided below.
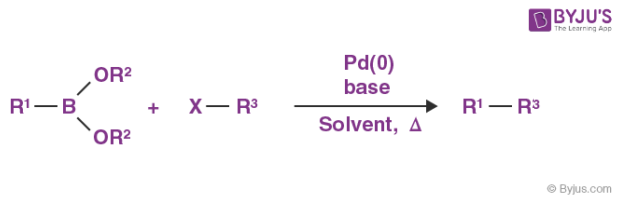
Here, R1 and R3 denote alkyl, alkene, or alkyne groups and R2 denotes an H-group or an alkyl group. Note that X denotes a halide group.
Types of Coupling Reactions
Coupling reactions can be classified into two types based on the chemical species that are combined by them:
- Homo-coupling reactions: Here, two identical chemical species are combined to yield a single product.
- Hetero-coupling reactions (also known as cross-coupling reactions): Here, two dissimilar chemical species are joined together to afford a single product.
Examples of Coupling Reactions
Examples of Homo-Coupling Reactions
An important example of homo-coupling reactions is the Ullmann reaction, which is illustrated below.
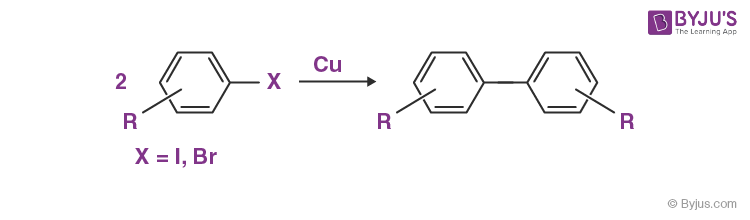
Other notable examples of such coupling reactions include:
- The Wurtz reaction
- The Pinacol coupling reaction
- Glaser coupling
Examples of Cross-Coupling Reactions
The Heck reaction is an important example of a cross-coupling reaction. The Heck reaction of an aryl halide and an alkene is illustrated below.
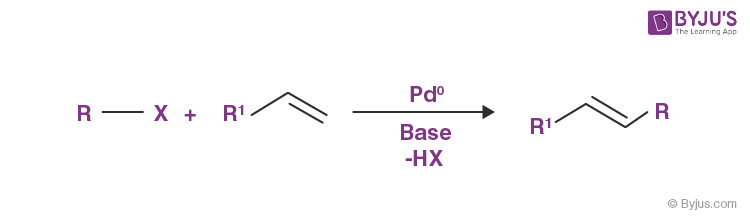
Other notable examples of cross-coupling reactions include:
- The Grignard reaction
- Sonagashira cross-coupling
- Suzuki coupling
- The Buchwald-Hartwig reaction
Applications of Coupling Reactions
Some important applications of coupling reactions are listed below.
- Coupling reactions are employed in the synthesis of several pharmaceutical products.
- Many conjugated polymers are prepared via coupling reactions that involve the use of a metal catalyst.
- The Suzuki coupling reaction (which is an important class of coupling reactions) is also used in the synthetic production of complex compounds. For example, it is used in the production of caparratriene, which is highly effective in the treatment of leukaemia.
- Many natural products can be obtained via certain coupling reactions. For example, benzylisoquinoline alkaloids can be synthesized with the help of the Sonogashira cross-coupling reaction.
Frequently Asked Questions – FAQs
What is coupling reaction in chemistry?
For a number of reactions where two fragments are joined together with the aid of a metal catalyst, a coupling reaction in organic chemistry is a general term. Hetero couplings combine two distinct partners, such as an alkene (RC = CH) and an alkyl halide (R’-X) in the Heck reaction, to give a substituted alkene.
What is the function of coupling?
Both couplings have the basic purpose of transmitting power, accommodating misalignment and compensating for axial movement (end shaft movement). A coupling is often requested to withstand shock or vibration.
What is the coupling mechanism the body uses?
Cells use a technique called reaction pairing in most situations, in which an energetically favourable reaction (such as ATP hydrolysis) is immediately related to an energetically unfavourable reaction (endergonic).
What is meant by a condensation reaction?
Condensation reaction, any of the groups of reactions in which, typically in the presence of a catalyst, two molecules interact with the removal of water or some other basic molecule. Self-condensation is defined as the synthesis of two similar molecules.
Why are coupled reactions important?
The hydrolysis of an ATP molecule leads to the dissolution of high-energy phosphate bonds, causing the exergonic form to release high quantities of energy. In order to transform this energy into an endergonic form, the coupled response plays an essential role. The electricity, then, is not extracted by the machine as heat.
To learn more about coupling reactions and other important types of organic reactions, register with BYJU’S and download the mobile application on your smartphone.

Comments