What is Osmosis?
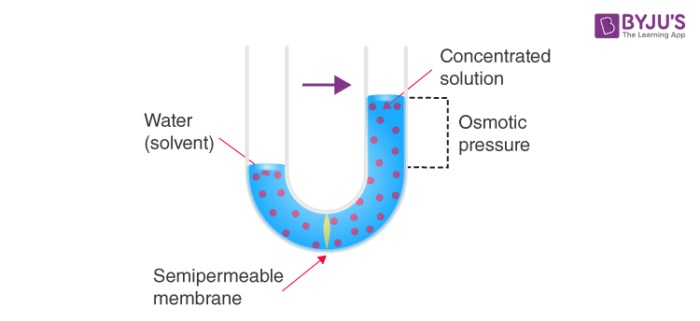
Osmosis is the movement of solvent molecules from the region of pure solvent (area of low solute concentration) towards the solution (area of higher solute concentration) through a semipermeable membrane.
Table of Contents
Osmosis is vital for many biological processes. Using the concept of osmosis and osmotic pressure, there is another process that was devised and is actively used in the purification of water. This process is called reverse osmosis (RO). You might have heard of the RO water purifiers being largely sold in the market. In this section, we will discuss reverse osmosis and how the process helps in the purification of water for drinking.
What is Reverse Osmosis (RO)?
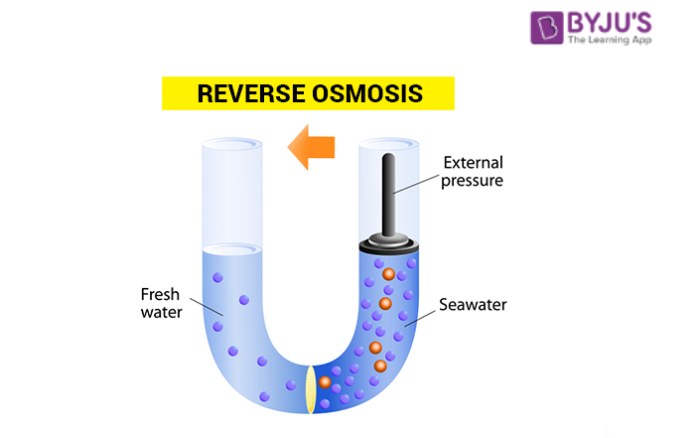
Reverse osmosis (RO) is a process in which a large pressure is applied to the solution side so as to overcome the osmotic pressure.
This pushes the pure solvent under pressure, out of the solution through the semi-permeable membrane. This process finds a number of practical applications. Some of them are the purification of drinking water, removal of salt from water molecules, removal of effluents from water, etc.
Recommended Videos

Water Purification
RO is considered as one of the most important water purification technologies. The contaminants present in water are removed by pushing water under pressure through a semi-permeable membrane. Pure water is thus squeezed out which is fit for drinking. RO is also used in the desalination of seawater. RO is a reliable source for obtaining potable water.
- The process eliminates dissolved and suspended impurities, even bacteria.
- The membrane is small enough to allow the solvent molecules to pass but does not allow large ions or molecules to pass through its pores.
- Polymer membranes are used for the desalinization of seawater.
- Cellulose acetate is often used as a semi-permeable membrane. It allows water molecules to pass but is impermeable to impurities.
- It finds application in the military, wastewater purification, food industry, landfill leachate purification, etc.
Check ⇒ Reverse Osmosis Experiment
To learn more about reverse osmosis, its applications and water purification techniques download BYJU’S The Learning App.

Comments