Introduction: Mixture
When two or more elements or compounds mix together, not necessarily in a definite ratio and do not interact chemically, then the resulting substance is known as a mixture. The components of the mixture have their own physical properties. For example a mixture of sand and water maintains the properties of both sand water. There are two types of mixtures: Homogenous and Heterogeneous. A homogenous mixture is a type of mixture in which the components mixed are uniformly distributed throughout the mixture and heterogeneous mixture is a type of mixture in which all the components are unevenly mixed. In this section we shall learn about the separation of mixtures.
Methods of separation of mixtures
The process of separating the inorganic substances present in the mixture or the constituents of the mixture by physical methods is known as separation of mixtures.
Chromatography
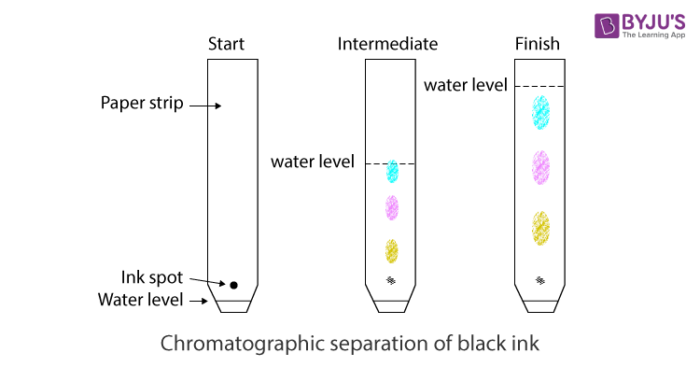
Chromatography is a method used to separate the different components in a liquid mixture. This method is based on the different properties of compounds in two phases: stationary and mobile phase. Based on this, chromatography can be classified into:
- Paper chromatography
- Column chromatography
- Thin layer chromatography
- Gas chromatography
Example: Separation of colours in dye
Centrifugation
This method is used to separate out tiny solid particles that usually pass through a filter paper and hence the separation of these insoluble particles is carried out with the help of centrifugation. The centrifugation process is based on the shape and size of particles, viscosity of the medium and speed of rotation. With the help of this process we can separate butter from cream. We use an apparatus known as centrifuge and this contains a centrifuge tube holder which contains equal amount of solid-liquid mixture. On rotating the rotor, due to the centrifugal force the denser insoluble particles separate from the liquid and ends up at the bottom of the tube and the liquid gets collected at the top.
To follow more about different methods of separation of mixtures, download BYJU’S – the learning app.

what are the methods of separation
Some of the common methods of separating substances or mixtures are:
Handpicking
Threshing
Winnowing
Sieving
Evaporation
Distillation
Filtration or Sedimentation
Separating Funnel
Magnetic Separation
Click here to learn about the Methods of Separation