What is Sodium Hydroxide?
Sodium Hydroxide is a solid ionic compound. It is also known as caustic soda, Iye, sodium hydrate or soda lye.
Sodium hydroxide is produced as a co-product in the production of chlorine. In its pure form, it is crystalline solid, and colourless in nature. This compound is highly water-soluble and consists of sodium cations and hydroxide anions. NaOH absorbs moisture from the air. It is highly corrosive and can cause severe skin burns and irritation to the eyes and other body parts.
It generates a high level of heat and so is always created by mixing the compound into the water, not vice versa. In cosmetics, this inorganic compound is used as a buffering agent. It can also control the pH levels. The pH of sodium hydroxide is 13. Sodium hydroxide is sometimes known as caustic soda. It’s a frequent ingredient in cleaning products and soaps. Sodium hydroxide is a white, odourless substance at room temperature. Sodium hydroxide liquid is colourless and odourless. It is extremely reactive to strong acids and water.
Table of Content
- Structure of Sodium Hydroxide
- Properties of sodium hydroxide
- Recommended Videos on Sodium hydroxide
- Preparation of sodium hydroxide
- Uses of sodium hydroxide
- Health Hazards of sodium hydroxide
- Safety Measures
- FAQs
Sodium Hydroxide Structure – NaOH
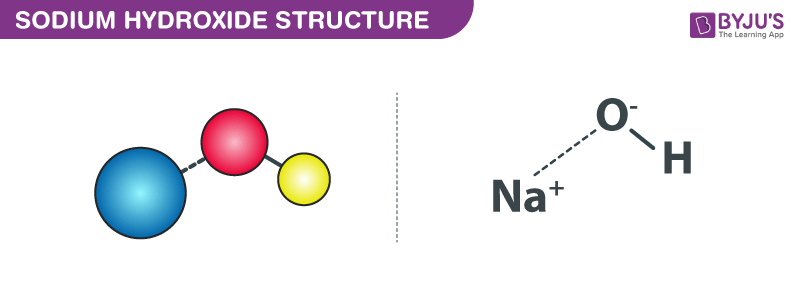
Sodium Hydroxide Structure – NaOH
Properties of Sodium Hydroxide – NaOH
Sodium Hydroxide is a white, translucent crystalline solid. It is commonly referred to as caustic soda due to its corrosive action on many substances; it decomposes proteins at room temperatures and may cause chemical burns to human bodies. Although it does not occur naturally, sodium hydroxide has been manufactured on a large scale for many years from readily obtainable raw material and is used in numerous industrial processes.
| NaOH | Sodium Hydroxide |
| Molecular Weight/ Molar Mass | 39.997 g/mol |
| Density | 2.13 g/cm³ |
| Boiling Point | 1,388 °C |
| Melting Point | 318 °C |
Recommended Videos
Why is Sodium Never Stored in Water?

Uses of Acids and Bases

Preparation of Sodium Hydroxide

Preparation of Sodium Hydroxide – NaOH
On a commercial scale, Caustic soda (sodium hydroxide) can be prepared by an aqueous solution of sodium carbonate (Na2CO3) is treated with hot milk of lime i.e Ca(OH)2 in a tank made up of iron.
Na2CO3 + Ca(OH)2 → CaCO3 + 2NaOH
(ppt.)
The precipitate of Calcium carbonate (CaCO3 ) is removed by filtration and the solution is used for paper making, soap and detergents.
Uses of Sodium Hydroxide- NaOH
- It is used in the manufacturing of detergents and soaps.
- It is used in the production of bleach-like chlorine.
- It is used in drain cleaners.
- It is used in the removal of heavy metals from the water by the municipal water treatment facility.
- It is used in food preservatives to prevent bacteria and mould growth.
- It is used for canning.
- It is used in papermaking and paper recycling.
Health Hazards of Sodium Hydroxide
- Contact with extremely high concentrations of sodium hydroxide can result in severe burns to the eyes, skin, digestive system, or lungs, which can lead to permanent damage or death.
- Mucous membranes in the nose, throat, lungs and bronchial system may be damaged. Even tiny doses can cause significant harm.
- Sodium hydroxide burns the skin and damages the eyes. The respiratory tract is irritated. Irritation occurs in the mucous membranes of the nose.
Safety Measures
1. Avoid coming into touch with your eyes, skin, or clothes.
2. Gases, fumes, dust, mist, vapour, and aerosols should not be inhaled.
3. Wear safety glasses, gloves, and clothes.
4. Acids should not be mixed. Do not eat, drink, smoke, or use personal products when handling chemical substances.
Frequently Asked Questions-FAQs
Why is sodium hydroxide toxic?
Humans can be exposed during the manufacture of sodium hydroxide and in the handling of sodium hydroxide as a solid or concentrated solution. Sodium hydroxide is corrosive to all body tissues; concentrated vapors cause serious damage to the eyes and respiratory system.
Is sodium hydroxide a salt?
Caustic soda (sodium hydroxide) is an alkali salt which is also called Lye. It is the common name of sodium hydroxide. This name is given due to the corrosive nature of this salt on animal and plant tissues.
What is the sodium hydroxide solution used for?
Sodium hydroxide is used in the manufacture of soaps and a range of detergents used in homes and business applications. NaOH is used for the production of bleach.
Is sodium hydroxide good for your skin?
Concentrated sodium hydroxide, when swallowed, is a strong irritant and corrosive to the skin, hair, breathing tract and digestive system. Sodium hydroxide has been combined with other components to create an effective, safe and versatile cleaning agent known as soap. Thus, sodium hydroxide is not good for our skin.
What is the importance of sodium hydroxide?
A metallic caustic base is sodium hydroxide (NaOH), also known as lye or caustic soda. An alkali, caustic soda is commonly used in many industries, mostly in the manufacture of pulp and paper, textiles, drinking water, and detergents as a strong chemical base.
Is sodium hydroxide soluble in ethanol?
Pure sodium hydroxide is a colourless crystalline solid which melts without decomposition at 318 ° C (604 ° F) and boils at 1,388 ° C (2,530 ° F). Despite lower solubility in polar solvents such as ethanol and methanol, it is highly soluble in water.
Why is sodium hydroxide used in soap?
It is not used for shaving, it is used for soap production. Oil and fat triglycerides react with sodium hydroxide to form fatty acid glycerol and sodium salts, the latter being the soaps. The extraction of excess sodium hydroxide is done with great caution because it is very caustic.
Also, Read:
| Bleaching Powder and Sodium Hydroxide | Uses of Sodium Hydroxide |
| Sodium | Preparation, Properties And Uses Of Sodium Chloride |
Learn more about the Structure and properties of NaOH from the experts at BYJU’S.

Comments