Glycerol formula is given here along with its structural and extended chemical formula. Glycerol is also known as glycerine or glycerine is a non-toxic, viscous, colourless, and simple triol compound. Glycerin or glycerol has several benefits and is mostly used in dermatological treatments and in the food industry as a preservative and sweetener.
Table of Content
Glycerol Chemical Formula
- Glycerol has 3 carbon atoms, 8 hydrogen atoms, and 3 oxygen atoms.
- It has a molecular weight of 92.09 g/mol and its IUPAC name is 1, 2, 3- Propanetriol or 1, 2, 3- Trihydroxypropane.
- The chemical and molecular formula for glycerol (glycerine) is given as-
| Chemical Formula of Glycerol | C3H8O3 |
| Extended Glycerol Molecular Formula | CH2OH-CHOH-CH2OH |
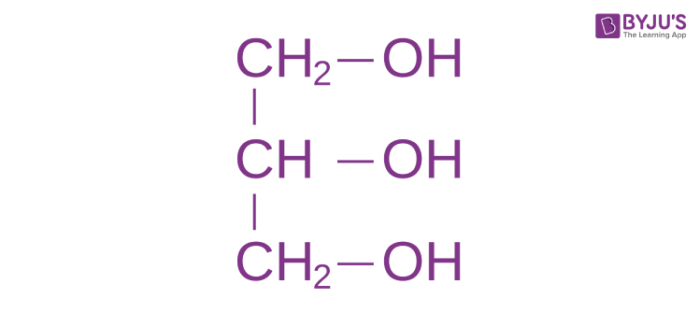
Structural Formula of Glycerol
- Glycerol molecule consists of a 3-carbon chain with 3 hydroxyl groups (OH) attached to each of them.
- The carbon atoms have sp3 configuration and so, the molecule has a free rotation. The glycerin structural formula is represented as-
- Glycerol is soluble in water and is hygroscopic.
- It is an important compound and its backbone is found in all lipids known as triglycerides.
- Its use ranges from pharmaceuticals to the food industry and in cosmetics (like soaps).
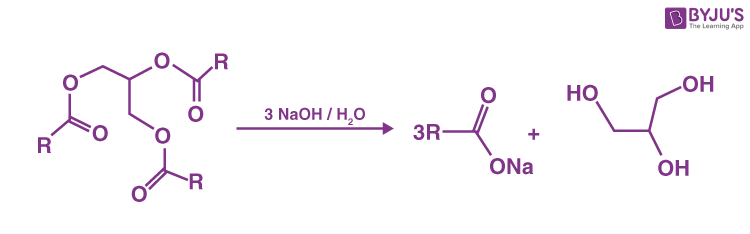
Production of glycerol
The triglycerides undergo hydrolysis, saponification, or transesterification producing glycerol as well as the fatty acid derivative. The crude glycerol form in this process is further purified by active charcoal to remove organic impurities, alkali to remove unreacted glycerol esters, and ion exchange to remove salts.
Uses of glycerol
- Glycerol is used to power diesel generators supplying electricity.
- It is used by the film industry when filming scenes involving water to stop areas from drying out too quickly.
- Glycerol is used as a replacement for water in ultrasonic testing.
- It is used to produce hydrogen gas.
To learn more about glycerol visit : Glycerin
To learn more chemical formulas of different compounds, stay tuned with BYJU’S.

Comments