What is Conformation of Cyclohexane?
Cyclohexane has a non-polar structure that makes it almost free from ring strain. The most important conformations that it can have included chain conformation and boat conformation. The chair conformation is more stable than the boat conformation. The boat conformation can sometimes be more stable than it is usually, by a slight rotation in the C-C bonds and is called the skew boat conformation. Nevertheless, the chair conformation is the most stable cyclohexane form.
A Conformation of cyclohexane can refer to many 3-Dimensional shapes assumed by a cyclohexane molecule without disturbing the integrity of the chemical bonds in it.
Table of Contents
Conformations of cyclohexane
A regular hexagon shape contains internal angles of 120o. However, the carbon-carbon bonds belonging to the cyclohexane ring have a tetrahedral symmetry, with the bond angles corresponding to 109.5o.
This is the reason why the cyclohexane ring has a tendency to take up several warped conformations (so that the bond angles are brought closer to the tetrahedral angle (109.5o) and there is reduced overall strain energy).
Examples of common conformations of cyclohexane include the boat, the twist-boat, the chair, and the half-chair conformations, which are named based on the shape that the cyclohexane molecule assumes in them.
These four cyclohexane conformations have been illustrated below along with some insight into their stability.
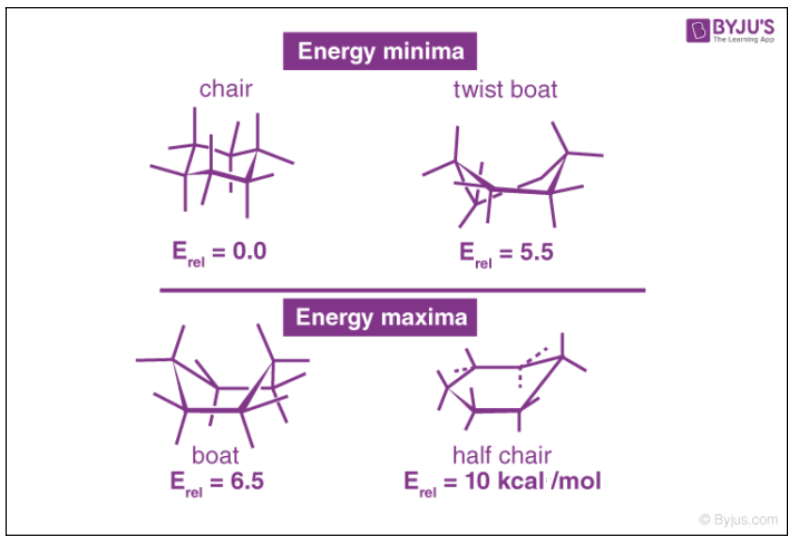
It can be noted that the cyclohexane molecule has the ability to switch between the conformations listed above and that only the chair and the twist-boat conformations can be isolated into their respective pure forms.
Due to hydrogen-hydrogen interactions in these conformations, the bond length and the bond angle vary slightly from their nominal values.
The chair conformations of cyclohexane have lower energies than the boat forms. However, the rather unstable boat forms of cyclohexane undergo rapid deformation to give twist-boat forms which are the local minima corresponding to the total energy.
The hydrogen atoms belonging to the carbon-hydrogen bonds that are at a perpendicular angle to the mean plane are called axial hydrogens, whereas those belonging to the carbon-hydrogen bonds which are parallel to the mean plane are called equatorial hydrogens. These bonds are also referred to as axial and equatorial bonds respectively.
Boat and Chair Forms of Cyclohexane
Cyclohexane is the most widely occurring ring in compounds of natural origin. Its prevalence, undoubtedly a consequence of its stability, makes it the most important of the cycloalkanes. The deviation of bond angle in cyclohexane molecules is more than in cyclopentane, it should be more strained and less reactive than cyclopentane. But actually, it is less strained and more stable than cyclopentane.
In order to avoid the strain, cyclohexane does not exist as a planar molecule as expected. It exists as a puckered ring which is non-planar and the bond angles are close to tetrahedral bond angles. Two such puckered rings for cyclohexane are called the boat and chair conformations.
Stability of conformations of cyclohexane
Generally, in the chair-shaped conformation of cyclohexane, there are two carbon-hydrogen bonds of each of the following types:
-
-
-
- Axial ‘up’
- Axial ‘down’
- Equatorial ‘up’
- Equatorial ‘down’
-
-
This geometry of chair cyclohexane conformations is generally preserved when the hydrogen atoms are replaced by halogen atoms such as fluorine, chlorine, bromine, and iodine. The phenomenon wherein the cyclohexane molecule undergoes a conversion from one chair form to a different chair form is called chair flipping (or ring flipping).
An illustration detailing chair flipping is provided below.

When chair flipping occurs, axial carbon-hydrogen bonds become equatorial and the equatorial carbon-hydrogen bonds become axial. However, they retain the corresponding ‘up’ or ‘down’ positions.
It can be noted that at a temperature of 25o Celsius, 99.99% of the molecules belonging to a given cyclohexane solution would correspond to a chair-type conformation.
The boat conformation of cyclohexane is not a very stable form due to the torsional strain applied to the cyclohexane molecule. The stability of this form is further affected by steric interactions between the hydrogen atoms. Owing to these factors, these conformations are generally converted into twist-boat forms which have a lower torsional strain and steric strain in them.
These twist-boat conformations of cyclohexane are much more stable than their boat-shaped counterparts. This conformation has a concentration of less than 1% in a solution of cyclohexane at 25o. In order to increase the concentration of this conformation, the cyclohexane solution must be heated to 1073K and then cooled to 40K.
Recommended Videos

Frequently Asked Questions – FAQs
Which conformation of cyclohexane is chiral?
Cyclohexane conformation free of angle strain: chair conformation is achiral because it has a centre of symmetry because boat conformation is achiral because it has a plane of symmetry. Twist boat conformation is chiral, since there is no element of symmetry.
Which is the most stable conformation of cyclohexane?
The chair form shown to the right is the most stable conformation of cyclohexane. The C-C-C bonds are very similar to 109.5o, so they are almost free from angle pressure. It is also a completely staggered conformation and is, therefore, free of torsional stress.
Which conformation of cyclohexane is the least stable?
Boat conformation is the least stable, with the highest strength, has steric hindrance on carbon 1 and carbon 4 between the two equatorial hydrogens, and has torsional stress, as each bond almost fully ellipses other bonds in the Newman projection.
Are diastereomers optically active?
Optical activity is the ability of a substance to rotate the plane polarised light. Chirality is the necessary condition for optical activity. Diastereomers lack mirror symmetry and are optically active.
Which conformation is more stable, axial or equatorial?
Since axial bonds are parallel to each other, substituents larger than hydrogen typically suffer from greater steric crowding when axial rather than equatorial driven. Replaced cyclohexanes would therefore preferentially follow conformations in which the larger substituents assume an equatorial orientation.

Which among these conformations of cyclohexane is chiral???
The twist-boat conformation of cyclohexane is chiral.