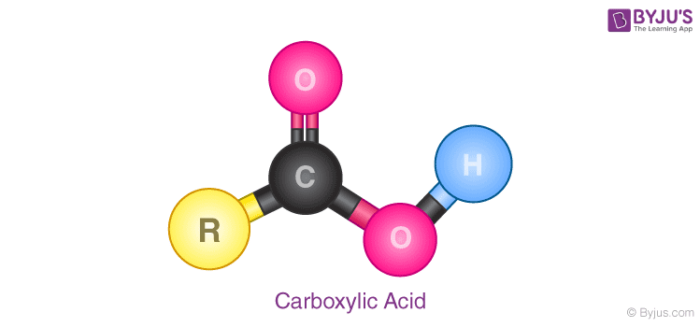
Carboxylic acid is a class of organic compounds that are characterised by the presence of the carboxyl group (-COOH) in them. The general formula for carboxylic acids is R – COOH where R refers to the rest of the molecule. The acidity of carboxylic acids is generally higher compared to simple phenols as they react with weak bases like carbonates and bicarbonates to liberate carbon dioxide gas.
Table of Contents
Carboxylates
Carboxylic acids are widely used, and they include amino acids and acetic acids. The salts and esters of carboxylic acids are known as carboxylates. When a carboxyl group is deprotonated, its conjugate base forms a carboxylate ion. The carboxylate ions are resonance stabilised and this increases their stability. This further leads to making them more acidic as compared to alcohol. There are also various methods that can be used for the preparation of Carboxylic acids.
However, carboxylic acids can be observed as a reduced form of Lewis-acid carbon dioxide, under certain conditions they can be decarboxylated to give carbon dioxide.
Uses of carboxylic acid
Carboxylic acids make up a series of fatty acids which are excellent for human health. Omega-6, as well as omega-3 fatty acids, are two examples. They help in maintaining the cell membrane and control nutrient use along with metabolism. If we consume a meal with unsaturated fat, the glucose and other nutrients will directly rush into the bloodstream without being absorbed. Whereas if there is an intake of saturated fat, digestion will slow down and the body will get more time to absorb the energy and nutrients from the meal.
The following points will state other significant uses of carboxylic acids:
- The manufacturing of soaps need higher fatty acids. Soaps are sodium or potassium salts of higher fatty acids such as stearic acid.
- The food industry uses many organic acids for the production of soft drinks, food products etc. For example, acetic acid is used in making vinegar. Sodium salts of organic acids find application in preservatives.
- In the pharmaceutical industry organic acids are used in many drugs such as aspirin, phenacetin etc.
- Acetic acids are often used as a coagulant in the manufacturing of rubber.
- Organic acids find huge applications in making dyestuff, perfumes and rayon.

Here you have learnt about the uses of carboxylic acids and if you are curious to know more, you can download and install BYJU’S – The learning app.

Comments