What is Urethane?
Urethane, also called ethyl carbamate is the ethyl ester of carbamic acid with the chemical formula C3H7NO2.
It has a broad spectrum of biological activities and is a known multisite carcinogen capable of inducing tumours in various animal species. In 1976 Ough was the first to demonstrate that ethyl carbamate was formed naturally in fermented foods such as bread, beer, soy sauce and wine.
Other name – Ethyl carbamate
Table of Contents
Properties of Urethane
| C3H7NO2 | Urethane |
| Density | 1.06 g/cm³ |
| Molecular Weight/ Molar Mass | 89.09 g/mol |
| Boiling Point | 182 to 185 °C |
| Melting Point | 46 to 50 °C |
| Chemical Formula | NH2COOC2H5 |
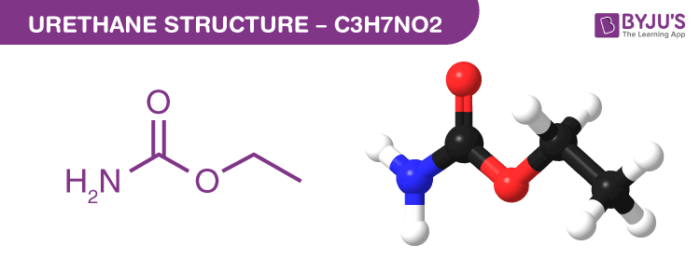
Urethane Structure – C3H7NO2
Physical Properties of Urethane – C3H7NO2
| Odour | no odour |
| Appearance | White solid |
| Complexity | 52.8 |
| Vapour Pressure | 0.36 torr at 25oC |
| Hydrogen Bond Donor | 1 |
| Solubility | readily soluble in water, alcohol and lipids. |
Chemical Properties of Urethane – C3H7NO2
- Urethane reacts with ethyl alcohol results in the formation of polyurethane and water. The chemical reaction is given below.
C3H7NO2 + C2H5OH → NH2CH2CH2COOC2H5 + H2O
- Urethane undergoes combustion reaction resulting in the formation of ammonia, carbon dioxide and water.
C3H7NO2 + 3O2 → NH3 + 3CO2 + 2H2O
Uses of Urethane – C3H7NO2
- Small amounts of ethyl carbamate were detected in fruit juices (9) that had been treated with an antimicrobial agent diethyl dicarbonate.
- Labelling experiments subsequently showed that urethane was formed from the reaction of DEDC with residual ammonia and that this mechanism could account for amounts found in orange juice, wine and beer.
- Used as chemical products, for example, pharmaceuticals, in biochemical research and medicine, and as pharmaceuticals in biochemical research and drug and as a solubilizer and co-dissolvable for pesticides and fumigants.
Frequently Asked Questions
What is urethane used for?
Urethane is a sealer used to secure many product forms but is most widely used to seal decorative concrete and stone. Some elements of polyurethane are also known as urethane. These sealers provide a barrier against fire, extreme cold, and moisture to protect the wood, concrete and other objects.
Is urethane safe?
Urethane is a potent carcinogen causing a wide variety of local and systemic tumours in laboratory animals. Urethane has been of low acute oral toxicity in mice. Following injection into rats and mice, adverse effects on the immune system occurred.
Is urethane rubber or plastic?
Polyurethanes are part of an artificial rubber family of elastomers. They’re exceptional in combining rigid plastics strength with rubber flexibility and elasticity. All-polyurethane applications include hoses, auto bushings, seat foam, skateboard frames, and spandex.
What is a urethane catalyst?
The amine-based urethane catalysts are used as traditional, reactive, delayed reaction and trimerization catalysts for polyurethane manufacturing. Urethane catalysts are used in industrial applications making paints, coatings, fibers, adhesives, and sealants.
Are urethane fumes harmful?
Yet avoiding polyurethane fumes may be the most important of all forms of fumes and toxins because of their potential for adverse side effects. When left unhealed, polyurethane can cause asthma and other problems with breathing.

Comments